HPLC, UHPLC
Published over 5 years ago. See the latest and most current information on HPLC, UHPLC.
In flight preparation pilots’ employ a rigorous systematic approach, using detailed checklists, due to the volume and detail of technical content in their pre or in-flight duties. People’s lives depend upon it.
Similarly setting up an (U)HPLC analysis is also very technical and people’s lives may depend upon the results of analysis. So why aren’t analysts as diligent with checklists? Standard operating procedures (SOP’s) are a form of checklist but rarely are these used in the same diligent manner as a pilot’s kneeboard checklist. Actually, most commercial airline pilots use electronic tablets for their checklists and only by selecting a response for each line item do they get to continue with the procedures and the next item to check.
Perhaps this points a possible direction for the separation science
and analytical world?
A very useful checklist that could be followed in the same vein as above is a systematic approach related to Troubleshooting HPLC retention time irreproducibility.
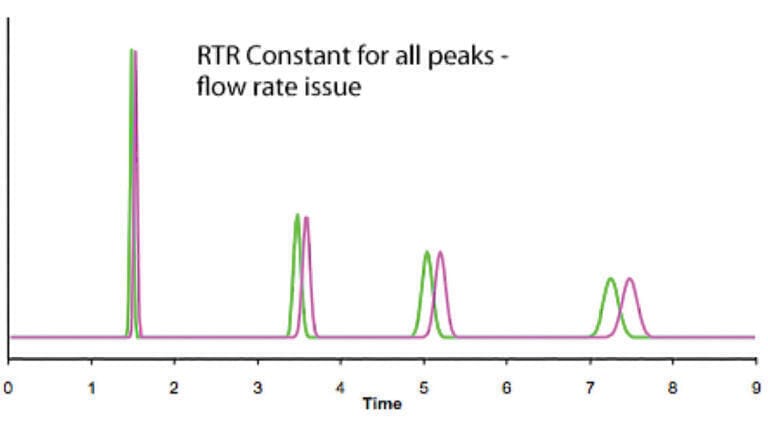
1. Measure the retention time change ratio for all peaks of interest:
Retention Time Retention Time Original Chromatogram
Change Ratio (RTR) = Retention Time Problem Chromatogram
2. Is RTR constant for all peaks?
a) Yes – the problem is likely to be a flow rate issue
b) Trending (increasingly large or small RTR value) – the problem may be related to %B solvent or a temperature change
c) No Trend (Random changes in RTR value) – selectivity change indicating pH, elution solvent or stationary phase issues (changes)
This is a very simple check list that most analysts will process in their minds, almost unknowingly, whilst evaluating troublesome data – even if RTR values are not computed, but very powerful in helping to diagnose the cause of problems when retention times shift.
There are several other ‘checks’ that we can run to generate confirmatory data.
The solvent void disturbance can be used to check and confirm whether an eluotropic or flow rate effect is occurring as is shown is the following examples.
3. Does the void marker of sequential analyses overlay or is it different?
Example 1: analyte retention time changes and void baseline disturbances also change – the issue is probably with a flow rate / eluent delivery problem.
Example 2: analyte retention time changes but void baseline disturbances overlay well – the issue is probably with the %B solvent, temperature or selectivity change.
Running evaluation tests using the peak area ratio (PAR) and peak height ratio (PHR) of the peaks of interest within the original and problem chromatograms to further confirm our diagnosis.
4. How does the PAR change?
Peak Area Change Ratio (PAR) = Peak Area Original Chromatogram
Peak Area Problem Chromatogram
a) All ratios change in constant proportion – suspect a flow rate issue or a problem with injector reproducibility. Of course, in the context of troubleshooting a retention time issue then flow rate would be the first consideration.
b) No change – suspect a problem with %B solvent or temperature
c) Random change – suspect a change in pH, eluent or stationary phase chemistry. There is a possibility that these random area ratio changes may be occurring due to adsorption of certain classes of analytes to active internal surfaces within the instrument, however, this should only be a consideration after checking the obvious variables of eluent pH, eluent and stationary phase chemistry.
5. How does the PHR change?
Peak Height Change Ratio (PHR) = Peak Height Original Chromatogram
Peak Height Problem Chromatogram
a) No change – suspect a flow rate issue
Flow rate reduced by 5%. Resulting in a retention and peak area change – peak height ratio (PHR) remains constant.
b) All ratios either increase or decrease but in an UNPREDICTABLE PATTERN – suspect a problem with %B solvent or temperature
%B solvent reduced by 3% Retention resulting in peak height changes not consistent.
c) Random change – suspect a change in pH / eluent or stationary phase chemistry
Mobile phase composition (selectivity) change resulting in random changes to retention time, peak area and peak height.
5. Here are some examples to further
explain some of these changes;
Why does area increase when flow decreases (for example)?
As flow rate slows the analyte moves through the detector flow cell more slowly, more data points are collected and the peak area increases. One should check the absolute peak areas, as any determination on Peak Area Ratio will likely remain constant.
For a concentration sensitive detector (absorbance, fluorescence), the concentration of analyte in the detector does not change, so the peak height does not change. The slight caveat is that if the flow rate change significantly affects the chromatographic efficiency, then as peak height is inversely proportional to peak width, any decrease in peak width (due to increased efficiency) will cause an increase in peak height and vice versa.
