Bioanalytical
Published over 9 years ago. See the latest and most current information on Bioanalytical.
On the 26th of May, 2016 the United Kingdom introduced the ‘Psychoactive Substances Act 2016’ [1], this law is intended to restrict the production, sale and supply of novel psychoactive substances (NPS) or as they are more commonly known, legal highs or designer drugs.
This law defines a ‘psychoactive substance’ as anything, which by stimulating or depressing the person’s central nervous system affects the persons mental functioning or emotional state.
In November 2016 Germany also introduced ‘The New Psychoactive Substances Act’ (NpSG) [2]. This law prohibits the acquisition, possession and sale of new psychoactive substances (NPS) as well as sanctioning the passing on of NPS. The NpSG specifically controls over two NPS groups; they are phenethylamines and synthetic cannabinoids.
As governments around the world make new laws to combat the production of the NPS, the effects of these substances are still real. Announcements in November 2016 on the ‘UNODC Early Warning Advisory on New Psychoactive Substances’ [3] documents increasing numbers of serious adverse events and deaths associated with several emerging synthetic opioids.
Authorities need to be able to rapidly detect the NPS with confidence and accuracy to tackle the problem. However, having the reference standard to confirm the presence of NPS is often difficult if the chromatography and analytical instrumentation such as a mass spectrometer rely on already knowing the details of specific compounds. This includes information such as retention time (RT), their mass to charge ratio (m/z) and the multiple reaction monitoring (MRM) transitions. Advanced systems that are accompanied by libraries which deliver high quality spectral data to confirm a known NPS, and contain these details (RT, m/z, MRM and libraries) are vital if you wish to conduct a screening assay using LC-MS/MS for example.
However, the NPS situation constantly evolves, as the clandestine laboratories that manufacture them make modifications to the structure (analogues), stereoisomers and derivatives. In doing this, the NPS may increase potency and alter the side effects of the designer drug. Once the NPS has been modified, it becomes difficult if not impossible to analyse.
In this study 85 analytes were selected which would challenge chromatographic separation and test the instrument to detect and screen the compounds in an ultra-fast time frame. Although in a real-life case it would be highly unlikely that any sample would contain this many forensic drugs.
In a recent application note ‘Ultra-Fast Forensic Toxicological Screening and Quantitation under 3 Minutes using Sciex X500R QTOF System and Sciex OS 1.0 Software’ [4] novel psychoactive substances were addressed in the study of urine samples. Twenty samples were diluted in 10% methanol and centrifuged to reduce the possibility of matrix interference. The clear supernatants were transferred to autosampler vials which were spiked with 85 compounds including opioids and synthetic cannabinoids. The urine samples contained a calibrator set spiked with compounds described in Table 1. There are 7 levels of calibrators: 40%, 80%, 100%, 200%, 300%, 500% and 1000% which are against the ng/mL value of each compound in table one, for example for the compound of 6-MAM, a metabolite of heroin, the calibration range was 4ng/mL to 100ng/mL. Using such a calibration range enable low level detection, sub cut off level through to a high level of concentration at 100ppb. There were also 20 urine samples with unknown number of compounds. The injection volume was 5 µL with a dilution factor for this sample set was a x4, again this reduces matrix interferences and the need for instrument maintenance.
Prior to the development of this method a 2-minute LC method [5] had been created using a Phenomenex Kinetex Phenyl-Hexyl column 50 × 2.1mm, 2.6µm 00B-4495-E0, however to improve the retention of the polar species the Hydro RP column was selected and the LC runtime was extended by 0.5 minute. Ensuring sufficient retention of the very polar species was important to allow the diversion of the salt-containing eluates in the beginning of the gradient to waste. The aim of refining the LC conditions was to evenly distribute all the eluting analytes throughout the duration of the data acquisition window to reduce the number of co-eluting analytes thus minimising ion suppression and matrix effects.
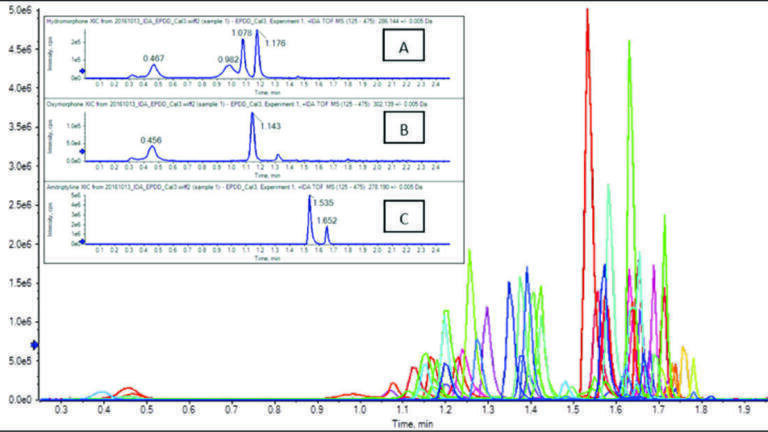
To preserve the column lifetime an organic wash was performed at the end of the gradient run followed by aqueous re-equilibration so retention time reproducibility was maintained. Within the 2.5 min LC runtime several groups of isomers could be resolved, in Figure 2 Morphine at 0.47 mins, hydromorphone at 0.98 mins, norcodeine at 1.08 mins were shown in Inset A, oxymorphone at 0.46 min and noroxycodone at 1.14 min were shown in Inset B, and amitriptyline at 1.54 min and EDDP at 1.65 min were shown in Inset C.
In this study the LC was coupled with the Sciex X500R QTOF system, a high-resolution quadrupole time-of-flight (QTOF) mass spectrometer. The X500R is suited for screening applications because the data generated from these systems provides structural information for every possible analyte. Typically, scanning across the full mass range provides the fine spectral details of the precursor ions of the analytes. After coupling to a quadrupole mass filter, such a mass spectrometer (Quadrupole-TOF or QTOF) can provide the full-scan information of not only the precursors ions, but also all the product ions in very high resolving power. Modern QTOF systems provide the capability of switching between MS and MS/MS scans instantly, enabling structural information to be obtained very quickly.
The analytical experiment to acquire the data was MS/MSAll with. In this technique’s data cycle, the instrument begins by acquiring the TOF-MS information first, and then sequentially follows that by acquiring the MS/MS information of all precursor ions across a specified mass range in pre-divided Q1 mass isolation windows. SWATH® acquisition records MS/MS information of everything all the time, and it significantly improves the MS/MS data quality by allowing sequentially programed Q1 isolations therefore more selective MS/MS data collection compared to other MS/MSAll techniques. Figure 3 shows the principal behind this acquisition experiment and Figure 4 details the settings for this acquisition on the X500R QTOF system.
Analysis of the data at spiking level 1 shows clear resolution and separation with confident identification. Figure 5 shows the extracted ion chromatograms of selected forensic analytes in urine at spiking level 1.
This analytical technique shows that even with a rapid LC gradient of less than 3 minutes, a full comprehensive profile of the sample can be built. Figure 6 shows the true positive rate at the varying concentration levels. Even at the low levels found in sample 1 there is still a 96% positive detection.
Unknown 1 and 2 were quality control urine samples spiked with all the analytes at different levels. Sample 3 through 20 were true unknown samples. There was a total of 82 true positive identifications from Sample 3 to Sample 20. For quantitation, the TOF-MS data from SWATH® acquisition can be used. It is worth mentioning that the MS/MS data from this acquisition can also be used for quantitation and compound identification through ion ratios, but we did not test that in this study but has been demonstrated in a previous study [6].
This full scan acquisition technique yielded excellent positive detection due to its complete coverage of MS/MS information. One of the key features of this scan type is the ability to create a digital archive of your sample, as this is an MSAll experiment of everything is scanned. The sample only needs to be injected once and the analysts will have a full insight as to what compounds are present in their sample, the analyst can return to this sample and re-interrogate the data at any time. There is no need to re-run a sample, which may have been subjected to degradation and thus forensic analytes may no longer be traceable. This means that if a brand new NPS enters the drug scene the acquisition will view the unique spectra attributed to the new forensic compound and begin to facilitate identifying the type of designer drug that has entered supply.
In this example, it has been demonstrated that tackling the chromatographic analysis of novel psychoactive substances with High Resolution Mass Spectrometry can be highly effective using an ultra-fast chromatographic separation technique coupled with a QTOF mass spectrometer. Non-targeted SWATH acquisition affords retrospective data analysis which is critically important for the rapidly emerging new designer drugs.
1. United Kingdom Psychoactive
Substances Act 2016 (c. 2) – ISBN 978-0-10-540028-8
2. Article 1 G. v 21/11/2016 . I S. 2615 ( # 55. ) Validity from 26.11.2016; FNA: 2121-12 Pharmaceutical and Medical Products Agency, poisons - New-Psychoactive Substances Act (NpSG) http://www.buzer.de/gesetz/12250/index.htm
3. Justice Tettey, S Chawla The challenge of new psychoactive substances – United Nations Office on Drugs and Crime - A Report from the Global SMART Programme March 2013 https://www.unodc.org/documents/scientific/NPS_Report.pdf
4. Xiang He, Adrian M. Taylor, Michael Jarvis and Alexandre Wang1
Ultra-Fast Forensic Toxicological Screening and Quantitation under 3 Minutes using SCIEX X500R QTOF System and SCIEX OS 1.0 Software. RUO-MKT-02-4931-A
5. Xiang He, Adrian M. Taylor – Forensic Identification and Quantification Workflows Delivered On A Revolutionary Designed QTOF And SCIEX OS Software. RUO-MKT- 02-3786-A.

