Bioanalytical
Published over 5 years ago. See the latest and most current information on Bioanalytical.
Next-generation antibodies such as multi-specifics, bi-specific T-cell engagers, peptibodies and nanobodies while offering greater therapeutic potential are more complex and diverse than traditional monoclonal antibodies. These new entities present significant analytical challenges, including the need to distinguish numerous structural and charge heterogeneities. Capillary zone electrophoresis enables the rapid separation of charge variants with only minor structural differences.
Development of new monoclonal antibody (mAb) drugs continues apace, but their limitations - monospecific binding sites and large size - has led researchers to explore next-generation antibody-based therapeutics that overcome these issues. Bi-specific and multi-specific mAbs, bi-specific T-cell engagers, peptibodies and nanobodies fulfill the requirements as some of them have multiple recognition sites, use only specific fragments of conventional antibodies thus providing greater in vivo stability, access to more targets and greater efficacy via multiple target binding.
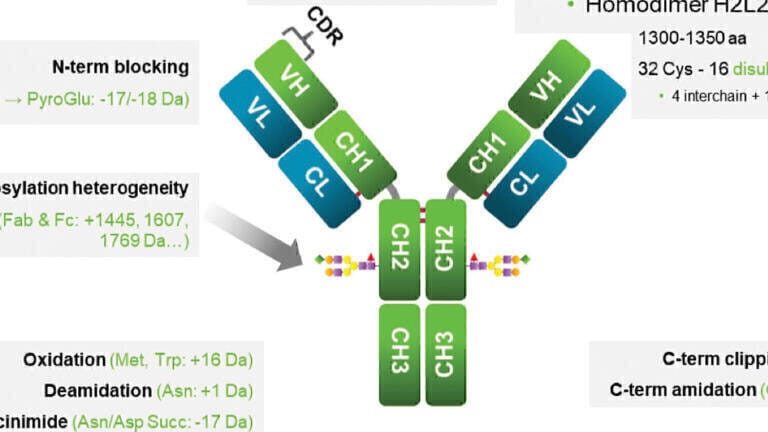
Some of these next-generation antibodies are similar to but more complex than immunoglobulin (IgG)-like mAbs with multiple Fab regions and one Fc region, such as bi- and tri-specific antibodies. Fusion proteins (single-chain variable fragments or scFvs), nanobodies, bispecific T-cell engagers (BiTEs), bi- and tri-specific killer-cell engagers (BiKes/TriKEs), and antibody fragments (Fab, F(ab)2) are non-IgG-like because they do not include an Fc region. The greater complexity of these next generation mAbs creates significant analytical challenges. Because they contain more components, numerous different but structurally similar variants are produced (see Figure 1), many of which may have the potential to impact safety and efficacy. These variants may arise as the result of deamidation, methionine oxidation, C-terminal lysine addition, N-terminal pyroglutamate formation and glycosylation, among other mechanisms [1-3].
Charge heterogeneity analysis of next-generation antibodies is consequently of importance during product development, production, stability and release testing [4]. Analysis of charge variants is also important during forced degradation studies for quality assessment [1]. Given that as more than a dozen variants can be generated in any one batch, only perhaps two of which will be therapeutically relevant, it is essential that any method for charge heterogeneity analysis be not only high resolution, but also rapid, accurate and reproducible. Traditional methods for protein charge variant analysis include ion exchange chromatography [5] and various forms of isoelectric focusing [6,7]. However, both chromatographic and IEF methods are relatively slow, requiring up to 1 hour of analysis time.
Capillary zone electrophoresis (CZE) has been used for charge heterogeneity analysis of protein therapeutics for more than a decade [8]. CZE operates by an entirely different mode of action than HPLC, i.e., separating analytes strictly based on the differential electromigration of the sample components based on their hydrodynamic volume to charge ratio (Figure 2, left panel).
With its non-laminar plug type flow profile, the analytes travel into narrow zones, and with no use of a stationary phase (e.g., no carryover), this method can be readily optimised for high-resolution separation of both large and small molecules with a wide range of chemical and physical properties (hydrophilic, hydrophobic, polar, non-polar, and charged) In fact, CZE offers plate numbers approximately several dozen times greater than that of high-performance liquid chromatography (HPLC) (Figure 2, right panel). In addition, CZE requires only nanolitre injection volumes and therefore minimises the consumption of high-value products. Furthermore, with minimal sample preparation requirements, this method represents a versatile separation platform for high-throughput screening of charge variants. If necessary, the electroosmotic flow can be minimised during capillary electrophoresis with the use of high-concentration zwitterionic buffer components such as epsilon amino caproic acid and specific capillary coating additives like triethylenetetramine [9].
In general, CZE enables analysis times of <10 minutes for next-generation protein therapeutics in their native forms. Studies have demonstrated excellent migration time and corrected peak area distribution reproducibility [10]. With minimal sample preparation for low-concentration samples (buffer exchange), CZE offers the ability for high-throughput analysis in all applications, including stability testing [11]. Figure 3 shows the charge variant analysis of the
NIST mAB by CZE with great reproducibility. In addition to the main peak, 5 smaller peaks were readily identified in less than 4 min
with the peak area distribution shown by the bar diagram.
To demonstrate the ultrafast charge variant analysis option of next-generation antibodies, the CZE Rapid Charge Variant Analysis Kit (SCIEX, Brea, CA) comprising CZE Rapid Charge Variant Separation Buffer, Acid Wash/Regenerating Solution, CE Grade Water and Protein Test Mix was used for the separation of the bispecific antibody of Emicizumab (Genentech, South San Francisco, CA) and the BiTE of Blinatumomab (Amgen, Thousand Oaks, CA), new generation drugs for the treatment of haemophilia A, and leukemia [12], respectively. CZE was performed on the PA800 Plus Pharmaceutical Analysis System (SCIEX) using the EZ-CE Pre-Assembled Capillary Cartridge (SCIEX), with all data acquired and processed by the 32 Karat version 10.1 software package (SCIEX).
The Emicizumab sample was prepared by diluting the bispecific antibody (30 mg/mL) with water to a concentration of 1 mg/mL. Six consecutive runs were completed (Figure 4), each in under 8 minutes, to reveal six peaks (inset in Figure 4). Peak 3 was the main component in the sample. Peaks 1 and 2 were faster migrating, possibly basic species, while peaks 4-6 were slower migrating, possibly acidic species.
The 6 consecutive runs for the Emicizumab sample resulted in the average migration time and corrected peak area percent reproducibility of 0.2 and 1.55 percent relative standard deviation (%RSD), respectively, as shown in Table 1.
The Blinatumomab sample contained a much lower concentration (0.2 mg/mL) of the BiTE active ingredient. Initially it was directly injected for CZE analysis, however, as can be seen in Trace A in Figure 5, no separation was achieved due to interference from the formulation buffer. Buffer exchange was attempted by washing the therapeutic protein sample three times with 100 μL of water through a 10 kDa cut-off centrifugation filter (Millipore, Billerica, MA) at 14 000 x g to eliminate these matrix-related components. The buffer-exchanged samples were subject to CZE analysis and resulted in the separation of five components in less than 6 minutes, as shown in Figure 5, Trace B. Peak 1 was a faster migrating, possibly basic ingredient, Peak 2 was the main component and Peaks 3-5 were slower migrating, possibly acidic species. The migration time and corrected peak area percent reproducibility were evaluated similarly to those discussed for the Emicizumab sample and determined to be 0.28% RSD and 1.58 % RSD, respectively
Many next-generation antibody compounds are produced at low concentration. Since each CZE run is typically <10 minutes in length, it is recommended for such low-concentration samples to first perform the analysis without any sample preparation. If insufficient separation is achieved, then simple buffer exchange via washing with water can be performed to eliminate the matrix effects.
Monitoring the deamidation of therapeutic proteins provides crucial information about the stability of the product. To show the applicability of rapid CZE analysis for such important stability studies, a three-day forced degradation of Emicizumab was performed at pH 8.7 and 45 ºC. The sample was prepared by diluting it with water and 1 M Tris-HCl buffer to obtain a 1 mg/mL protein solution in 100 mM Tris-HCl buffer and then incubated in a heating block. The samples were buffer exchanged on 10 kDa cut-off filters prior to analysis. Figure 6 depicts the resulting data for the separated species using CZE and cIEF modes. CZE is a rapid screening option that can reveal changes in concentration of different components within a sample during forced degradation studies. Because of its mode of action, however, it cannot be used to identify which peaks are acidic and which are basic. For such a determination, capillary isoelectric focusing (cIEF) method can be used. Albeit, cIEF runs take longer (typically take up to 30-40 minutes), it provides isoelectric point (PI) values. Figure 6 presents a comparison of the results obtained by using CZE and cIEF. It is clearly seen from this figure that the CZE technique can be readily applied to rapid, high throughout charge heterogeneity profiling. In the case of Emicizumab, CZE in fact provided results approximately five times faster than cIEF with apparently identical relative peak area for the main peak, 53% in both cases.
Capillary zone electrophoresis, with its unique mode of action, brings key benefits for charge variant analysis that are needed for novel antibody modalities but not provided by other methods.
CZE offers high-resolution, accurate, reproducible separation of structurally similar charge variants. High-concentration samples can simply be diluted with water prior to injection, while simple buffer exchange may be necessary to eliminate formulation matrix effects for low-concentration samples.
For different types of next-generation antibodies (a bispecific and a BiTE), separations were accomplished in <8 minutes with excellent migration time and corrected peak area percent distribution reproducibilities. In addition, CZE provided similar quantitative results to those obtained using the slower cIEF method when used to analyse samples collected during forced degradation studies.
As importantly, the CZE technique allows for true high-throughput screening and rapid analysis for development, in-process and product release testing applications. Overall, therefore, the CZE method described herein can be readily applied for high-throughput charge heterogeneity analysis of new therapeutic modalities.
It is also worth noting that capillary zone electrophoresis in combination with mass spectrometry enables the identification of modified peptides by separating them from their non-deamidated counterparts, which is not possible with LC due to the marginal mass differences of these molecules.
The technical help of Dr Zoltan Szabo is greatly appreciated.
1. Hsu, Y.R., et al., Selective deamidation of recombinant human stem cell factor during in vitro aging: isolation and characterization of the aspartyl and isoaspartyl homodimers and heterodimers. Biochemistry, 1998. 37(8): p.2251-62.
2. Harris, R.J., Processing of C-terminal lysine and arginine residues of proteins isolated from mammalian cell culture. J Chrom A, 1995. 705(1): p.129-34.
3. Moorhouse, K.G., et al., Validation of an HPLC method for the analysis of the charge heterogeneity of the recombinant monoclonal antibody IDEC-C2B8 after papain digestion. J Pharm Biomed Anal, 1997. 16(4): p. 593-603.
4. Singh, S.K., G. Narula, and A.S. Rathore, Should charge variants of monoclonal antibody therapeutics be considered critical quality attributes? Electrophoresis, 2016. 37(17-18): p.2338-46.
5. Stoll, D., et al., Characterization of therapeutic antibodies and related products by two-dimensional liquid chromatography coupled with UV absorbance and mass spectrometric detection. J Chrom B Anal Tech Biomed Life Sci, 2016. 1032: p.51-60.
6. Righetti, P.G., Determination of the isoelectric point of proteins by capillary isoelectric focusing. J Chrom A, 2004. 1037(1-2): p.491-9.
7. Sosic, Z., et al., Application of imaging capillary IEF for characterization and quantitative analysis of recombinant protein charge heterogeneity. Electrophoresis, 2008. 29(21): p.4368-76.
8. He, Y., et al., Analysis of identity, charge variants, and disulfide isomers of monoclonal antibodies with capillary zone electrophoresis in an uncoated capillary column. Anal Chem, 2010. 82(8): p.3222-30.
9. Moritz, B., et al., Evaluation of capillary zone electrophoresis for charge heterogeneity testing of monoclonal antibodies. J Chrom B Anal Tech Biomed Life Sci, 2015. 983-984: p.101-10.
10. Guttman, A. SCIEX Webinar, March 2021 (RUO-MKT-11-12884-A).
11. Filep, Cs. et al., Multilevel capillary gel electrophoresis characterization of new antibody modalities, Analytical Chimica Acta, 2021. 1161: In Press.
12. Szabo, Z and Guttman, A., Rapid charge heterogeneity analysis of new therapeutic protein modalities, SCIEX Technical Note, 2020 (RUO- MKT-02-12580-A).
