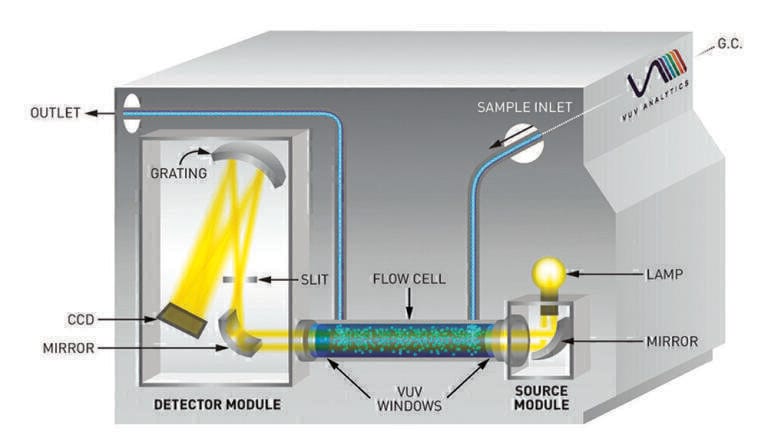
Gas chromatography
Published over 6 years ago. See the latest and most current information on Gas chromatography.
Non-traditional gasoline additives (NTGAs) are being researched as beneficial octane-booster replacements for ethanol and methyl tert-butyl ether. Other octane-enhancing NTGAs, including acetone and N-methylaniline which can degrade automobile engine performance, are sometimes used illegally in gasolines outside the United States and Europe. Unfortunately, monitoring programs relying on ASTM D6730 (detailed hydrocarbon analysis) and ASTM 6839 (multidimensional gas chromatography) do not include most NTGAs in their scope. Gas chromatography (GC) with vacuum ultraviolet spectroscopy (VUV) offers an easy way to spectrally identify and quantify NTGAs in gasoline in under 34 minutes.
1. Introduction:
During the fuel refining process, various additives are blended into the fuel stream to adjust the fuel’s properties. Properties of interest include vapour pressure [1,2], exhaust emission content [3], water tolerance [4], and corrosiveness [5], among others. Many of these additives are used in a range of concentrations, from bulk down to trace levels, depending on the compounds. One particular property affected by additive content is the octane rating, or octane number. The octane rating is a fuel’s ability to resist ‘knocking’ or ‘pinging’ as a result of premature combustion [6]. Modern vehicles are designed to operate with fuel at a specific octane rating; for example, most light duty cars and trucks are designed to use fuel with an octane rating of 87, which can be found at most gas stations [7].
Many ‘traditional’ additives, such as benzene, toluene, ethylbenzene, and xylenes (BTEX) have been used to boost the octane rating [8,9]. However, these compounds are heavily regulated, forcing oil refineries to search for renewable and environmentally-friendly additives. Over the last two decades, ethanol has become an increasingly popular additive, and it is now blended at approximately 10% by volume in many gasolines [8]. Ethanol has many desirable properties, such as high biodegradability, low toxicity, and efficient burning [10]. However, it contains less energy per gallon than gasoline [11], and the increased requirements for crops such as corn would drive up food prices [12], so using too much ethanol in gasoline is not desirable.
Recently, research has been dedicated to using new ‘non-traditional’ gasoline additives (NTGAs) as viable additions to fuel blends [7,13]. For example, one study determined that ethyl acetate may be used as a beneficial octane-boosting compound that provides desirable properties, such as increased water tolerance [1]. Another study demonstrated a procedure for obtaining various furan compounds from pineapple plantation waste residues which could be used in gasoline [14].
While many NTGAS are beneficial and used ethically, several harmful, illegal additives have been found in gasoline. The Asian Clean Fuels Association (ACFA) identified octane-boosting additives that have undesirable side effects. Compounds such as acetone and dimethoxymethane (methylal) can cause swelling of plastic engine components, potentially leading to engine damage. Other harmful NTGAs may have negative effects on gasoline, including volatility, gum formation, and corrosion [15].
As the content of gasoline changes with advancing fuels technology, gasoline regulatory procedures must incorporate proper analytical techniques to analyse NTGAs. Currently, gasoline is analysed using methods such as ASTM D6730 (detailed hydrocarbon analysis, or DHA) or ASTM D6839 (multidimensional gas chromatography) [16,17]; however, these methods don’t include most NTGAs within their scope. Due to the nature of these techniques, NTGAs may prove problematic. NTGAs in gasoline may coelute with known compounds in a DHA, but because DHA cannot provide spectral or structural information, quantitation may be affected [18]. Additionally, techniques like multidimensional GC may require changes in the analysis mode to properly analyse the NTGAs, increasing the complexity of the analysis [19].
A recently developed technique, gas chromatography - vacuum ultraviolet spectroscopy (GC-VUV), has proven to be a powerful alternative to traditional methods of gasoline analysis. Molecules eluting from the GC are exposed to light in the VUV range (125-240 nm), and because nearly every compound absorbs strongly in this range, compounds can be identified by their unique spectral fingerprints and quantified according to Beer-Lambert Law principles. Additionally, coeluting compounds can be distinguished by spectral deconvolution, allowing for additional chromatographic flexibility [20]. ASTM D8071 uses GC-VUV to analyse the paraffin/isoparaffin/olefin/naphthene/aromatic (PIONA) and oxygenate content of fuels samples in a single run, and because it uses a simple setup with a run time under 34 minutes, it is an ideal solution for fuels analysis [21].
This study aims to demonstrate GC-VUV’s ability to analyse gasoline samples containing NTGAs. Rather than tailoring the method conditions to the new compounds, information on each NTGA was simply added to the spectral library and the samples were analysed using the method conditions listed in ASTM D8071 [21].
2. Experimental:
All samples were analysed with an Agilent 6890 gas chromatograph (Wilmington, DE, USA) paired with a VUV Analytics VGA-100 spectrometer (Cedar Park, TX, USA). The GC was equipped with a Restek Rxi-1ms column (Bellefonte, PA, USA) with dimensions of 30m x 0.25 mm ID x 0.25 μm and a Restek Topaz 4mm ID Precision inlet liner with wool. Helium with a purity of 99.999% was used as the carrier gas for the GC. Nitrogen with a purity of 99.999% was used as the makeup gas for the spectrometer.
All samples were run under the conditions specified by ASTM D8071 as follows:
Gas chromatograph
• Constant flow mode: 1.0 mL/min He
• GC inlet: 250°C, split 300:1
• Injection volume: 1 µL
• Oven: 35°C (10 min), 7°C/min to 200°C
VGA-100
• Makeup gas: 0.25 psi N2
• Acquisition range: 125-240 nm
• Acquisition rate: 4.5 Hz
• Transfer line temperature: 275°C
• Detector flow cell: 275°C
Oxygenate-free gasoline was obtained from Valero (San Antonio, TX, USA), and E10 gasoline (containing 10% ethanol) was obtained from a local gas station. All NTGA compounds and other reagents were purchased from Sigma Aldrich (St. Louis, MO, USA) with a purity of >99%. Each compound analysed as an NTGA is listed in Table 1.
Individual solutions of each NTGA were prepared by dissolving each compound in oxygenate-free gasoline at concentrations of 10%, 5%, 3%, and 1% v/v and analysed in triplicate. One compound, gamma-valerolactone, was not soluble at 10% in oxygenate-free gasoline, so it was dissolved in E10 gasoline at concentrations of 10%, 5%, 3%, and 1% v/v and analysed in triplicate. Additionally, individual solutions of three compounds - dimethoxymethane, 2,5-dimethylfuran, and N-methylaniline - were prepared with concentrations of 20%, 10%, 5%, 2%, 1%, 0.5%, 0.2%, 0.1%, and 0.05% v/v in E10 gasoline and again analysed in triplicate.
All data was processed using retention indices (RI) relative to methane and spectral matching. Quantification was achieved by comparing each compound’s response to benzene and calculating a relative response factor (RRF). Procedures for determining RI and RRF values are listed in ASTM D8071 [21]. The RI and RRF values used in this experiment can be found in Table 2.
3. Results and Discussion:
The quantification results for each NTGA can be found in Table 3. Each compound of interest was quantified in gasoline over a range of 1% - 10% v/v.
While most NTGAs would likely fall between 1% and 10% v/v, this concentration range can be expanded. Three additives were chosen to be analysed over a range of 0.05% - 20% v/v and were analysed in triplicate. Those results can be found in Table 4.
Unlike methods such as DHA which rely on chromatographic separation to identify compounds through retention time, GC-VUV takes advantage of spectral deconvolution to compress the chromatography, giving a shorter run time of 34 minutes. While NTGAs may coelute with other compounds in gasoline, spectral deconvolution allows them to be identified and quantified without issue. For example, although acetone coelutes with isopentane under these conditions, it was quantified over the 1% - 10% v/v linear range with an R2 value of 0.998. An example of spectral deconvolution can be seen in Figure 2. The spectral differences between acetone and isopentane allowed them to be distinguished and characterised separately despite their similar retention indices.
4. Conclusions
GC-VUV is able to successfully analyse and quantify several NTGAs in a 34-minute run time. These NTGAs can be uniquely identified by their spectral fingerprints, and spectral deconvolution allows these compounds to be identified and quantified even if they coelute with other compounds in gasoline. Highly linear data can be obtained for each NTGA over a wide range of concentrations without sacrificing the PIONA and oxygenate analysis necessary for fuel stream monitoring and regulation. Additionally, the ability to analyse these compounds without changing the hardware configuration or method parameters greatly simplifies the analysis, making it a desirable alternative to cumbersome methods such as ASTM D6730 or ASTM D6839.
Despite its advantages, NTGA analysis with GC-VUV is limited in part by the VUV spectral library. Unlike mass spectral libraries such as the one from NIST, which have been built over multiple decades, the VUV spectral library is relatively small. It contains entries for a large number of gasoline compounds and is continually growing, but many NTGAs may not exist in the library. If data is not available, unknown NTGA peaks may need to be identified with another technique, such as GC-MS. Once identified, a reference standard of the compound must be purchased and analysed so it can be included in the spectral library. However, once the compound is in the library, it can be analysed and quantified using GC-VUV as described in this paper.
5. References:
1. M. Amine, M.A.H. Zahran, E.N. Awad, S.M.El-Zein, Y. Barakat. International Journal of Modern Organic Chemistry 2(3) (2013) 226-250.
2. United States Environmental Protection Agency. Gasoline Reid Vapor Pressure (2018). https://www.epa.gov/gasoline-standards gasoline-reid-vapor-pressure#information.
3. United States Environmental Protection Agency. Final Rule for Model Year 2017 and Later Light-Duty Vehicle Greenhouse Gas Emissions and Corporate Average Fuel Economy Standards (2018). https://www.epa.gov/regulations-emissions-vehicles-and-enginesfinal-rule-model-year-2017-and-later-light-duty-vehicle
4. Renewable Fuels Association. E15 Retailer Handbook (2013). https://www.epa.gov/sites/production/files/2016-06/documents/rfae15-retailer-handbook.pdf.
5. United States Environmental Protection Agency. Emerging Fuels and Underground Storage Tanks (USTs) (2018). https://www.epagov/ust/emerging-fuels-and-underground-storage-tanks-usts.
6. United States Department of Energy. Selecting the Right Octane Fuel. https://www.fueleconomy.gov/feg/octane.shtml.
7. J. Yanowitz, E. Christensen, R.L. McCormick. Utilization of Renewable Oxygenates as Gasoline Blending Components (2011). National Renewable Research Laboratory. https://www.nrel.govdocs/fy11osti/50791.pdf.
8. J. Stolark. Fact Sheet – A Brief History of Octane in Gasoline: From Lead to Ethanol (2016). Environmental and Energy Study Institute. https://www.eesi.org/papers/view/fact-sheet-a-brief-history-of-octane#3.
9. BP. Fuel News: Fuel, Octane & Power (2010). https://www.bp.com content/dam/bp-country/en_au/media/fuel-news/fuel-octane-power.pdf.
10. United States Energy Information Administration. Biofuels: Ethanol and Biodiesel. https://www.eia.gov/energyexplainedindex.php?page=biofuel_ethanol_environment.
11. United States Department of Energy. Ethanol Benefits and Considerations. https://afdc.energy.gov/fuels/ethanol_benefits.html.
12. Congressional Budget Office. The Impact of Ethanol Use on Food Prices and Greenhouse-Gas Emissions (2009). http://largestanford.edu/courses/2011/ph240/sojka2/docs/04-08-Ethanol.pdf.
13. R.L. McCormick, G. Fioroni, L. Fouts, E. Christensen, J. Yanowitz, E. Polikarpov, K. Albrecht, D. J. Gaspar, J. Gladden, A. George. SAE International Journal of Fuels and lubricants 10(2) (2017). https://www.osti.gov/pages/servlets/purl/1378889.
14. A.P. Irías-Mata, G. Lutz. Cuadernos de Investigación UNED 5(2) (2013), 279-282.
15. Asian Clean Fuels Association. Report on Harmful Chemicals in Gasoline Blending (2014). https://www.acfa.org.sg/pdf InFocus17_2014_10_Report_on_harmful_chemicals.pdf.
16. ASTM International. ASTM D6730, Standard Test Method for Determination of Individual Components in Spark Ignition Engine Fuels by 100–Metre Capillary (with Precolumn) High-Resolution Gas Chromatography (2016). https://www.astm.org/StandardsD6730.htm.
17. ASTM International. ASTM D6839, Standard Test Method for Hydrocarbon Types, Oxygenated Compounds, and Benzene in Spark Ignition Engine Fuels by Gas Chromatography (2018). https://www.astm.org/Standards/D6839.htm.
18. California Environmental Protection Agency. SOP No. MLD 118: Procedure for the Detailed Hydrocarbon Analysis of Gasolines by Single Column High Efficiency (Capillary) Column Gas Chromatography (1997). https://www.arb.ca.gov/testmethslb/slb118.pdf.
19. Bruker Daltonics Inc. Application Note CA703979: Fast Gasoline Characterization by Optimizing Multi-dimensional GC (PIONA+). https://www.bruker.com/fileadmin/user_upload/8PDF-Docs/Separations_MassSpectrometry/Literature/literatureApplicationNotes/PIONA_AppNote_CA703979.pdf.
20. K.A. Schug, I. Sawicki, D.D. Carlton Jr., H. Fan, H.M. McNair, J.P. Nimmo, P. Kroll, J. Smuts, P. Walsh, D. Harrison. Anal. Chem. 86 (2014), 8329-8335.
21. ASTM International. ASTM D8071, Determination of Hydrocarbon Group Types and Select Hydrocarbon and Oxygenate Compounds in Automotive Spark-Ignition Engine Fuel Using Gas Chromatography with Vacuum Ultraviolet Absorption Spectroscopy Detection (GC-VUV) (2017). https://www.astm.orgStandards/D8071.htm.

