Help Desk
Published over 7 years ago. See the latest and most current information on Help Desk.
The pharmaceutical industry is seeing a shift in the types of therapeutic molecules that it produces. Although there is still growth in the low-molecular-weight substance for active pharmaceutical ingredients, the greatest growth is in the development of biotherapeutics. With the major area of focus being the development of protein-based monoclonal antibodies such as Adalimumab, Rituximab, infliximab, trastuzumab and bevacizumab which are currently in the top ten list [1] of drug sale revenues across all genres of drugs. Qualitative and quantitative determination of proteins and any impurities manufactured during the biosynthetic production process is therefore an important consideration when looking at the pharmaceutical drug development cycle. The current approach uses a variety of chromatographic tools to realise the required information, with one of the most important characterisation techniques employed being peptide mapping. However, there are challenges with the chromatographic separation of peptides and the HelpDesk will review some of these challenges relating to the choice of modifier.
Proteins are highly complex molecules which are typically defined by three layers of structure, Figure 1;
• Primary – amino acid sequence
• Secondary – short range structure due to the formation of intra molecular bonds between different parts of the polypeptide chain. The bonds formed are typically hydrogen bonds and result in the formation of α-helix and β-sheets.
• Tertiary – Associated with the formation of longer range interactions between different amino acids groups within the polypeptide chain. The interactions would include, ionic, hydrophobic, electrostatic interactions and formation of disulphide bridges.
Figure 1: Schematic diagram of protein showing three layers of structure
Understanding the primary structure of the protein and the fragmentation of the protein into peptides are important consideration. This is typically achieved by using a specific enzyme which has exceptional cleavage specificity, cleaving proteins on the carboxyl terminal side of arginine and lysine amino acid residues, except when either is followed by proline. This results in the generation of peptides with an average size of 700 to 1500 Daltons, which is ideal for LC-MS. The process of producing signature peptides is substantially more involved than just adding the trypsin protease to the native protein, however the focus of this article is the resulting peptide separation, and how this can be optimised.
Peptide mapping, which is the analysis of the final peptide mixture is typically performed using reversed phase chromatography. Assuming that the enzymatic digestion has been performed well, this approach will allow for a complete identification of the amino acid sequence for a specific protein when coupled to MS detection. Traditionally, when using UV detection this separation has been performed with the use of trifluoroacetic acid (TFA) as an additive in the mobile phase. The TFA is added to reduce the amount of tailing associated with the interactions of the peptides with the stationary phase, predominantly residual silanols associated with the silica substrate. There are several aspects to consider here. The first is that reducing the pH will result in the less active forms of the silonyls becoming neutralised, and so reducing interactions with the basic moieties of the peptides. Also the TFA can act as an ion pairing reagent, interacting directly with the peptides, resulting in a neutral form of the peptide, which will also result in reducing the interactions with the silanol surface. Manufacturers have gained a much better understanding of these interactions which has resulted in the generation of less active substrate silicas. The use of TFA as a modifier is commonplace in the field of chromatography due to the properties explained previously. However, with the coupling of liquid chromatography to mass spectrometry the ion pairing results associated with reduction of sensitivity and as a result separation scientists are investigating other acids, such as formic acid, as possible replacements.
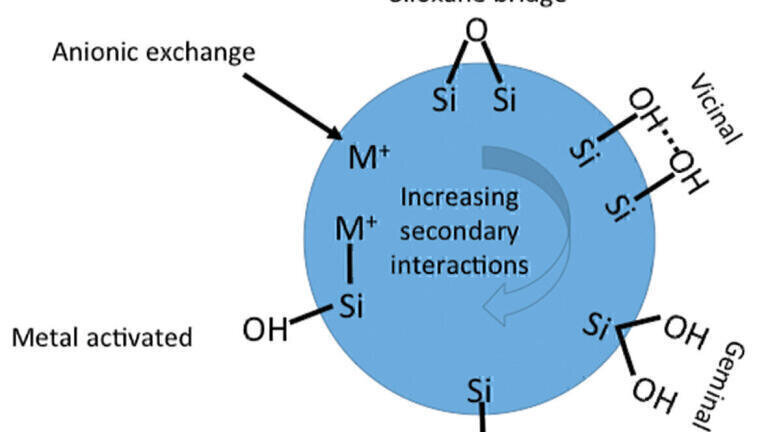
Different forms of silanols, having different physiochemical properties, are present on the surface of the silica substrate and as a consequence the surface is not homogeneous. Figure 2 shows schematically a few of these different forms of silanols that exist on the surface. The reactivity of the different substrate surface groups increases as shown in the diagram, with a low activity for the siloxane bridge, with the highest activity being derived from a metal activated silanol.
To understand the chromatography interactions taking place when these acidic mobile phase additives are employed, it is necessary to look at the dissociation constants of the surface silanols relative to these acids. When formic acid is used there will be a mixture of protonated and unprotonated sites on both species when using formic acid mobile phases. TFA is a stronger acid, having a lower pKa, so it dominates, giving up its proton and fully protonating and neutralising the silanols. Another approach to getting better peak shape is to remove the more acidic free silanols from the surface of the substrate, reducing the secondary interactions, resulting in less tailing. This can be achieved in a variety of manners, including blocking sites using endcapping reagents, or the use of higher purity silicas and thus reducing the more acidic forms of the silanols. This allows the use of the weaker formic acid as a mobile phase additive without the tailing that is associated with low purity silicas or silicas which are not effectively end-capped. Better peak shape aids both quantification and also identification when performing peptide analysis.
With more laboratories using MS detection for peptide mapping, columns that perform well with formic acid rather than TFA are gaining popularity. A common problem in liquid chromatography is peak tailing. This can cause issues with quantification and identification which results in a loss of robustness of an assay.
Column manufacturers go to great lengths to block these residual silanols however it is extremely difficult to do completely. At the moderate acidity (pH ~ 4) of formic acid mobile phases, these negatively charged silanols can have undesirable ionic interactions with positively charged proteins and peptides, leading to tailing and poor peak shape, Figure 3 and Figure 4.
TFA is a strong enough acid that these silanols are neutralised, the TFA anion forms ion pairs with positive charges on the peptide; the ionic interactions are blocked, and the resulting separation is based purely on reverse-phase interactions. Formic acid partially blocks these interactions but is not as effective as the stronger acid, Figure 5.
The use of TFA not only reduces tailing but can also affect the separation. Figure 6 shows the effect of altering the amount of TFA. Using a small amount of TFA results in a separation of previously co-eluting components and also a shift in retention of the major peaks. It can also be seen that there is a substantial reduction in the sensitivity, with the ion count being reduced from 106 to 3 .05.
Figure 6: The effect of using TFA as an additive in the mobile phase. TFA affects the selectivity and the sensitivity of the assay.
The use of mobile phase additives, when using LC-MS, can have a significant effect on the peak shape, separation and retention time of individual components as well as the sensitivity. Even small amounts of TFA can reduce the signal intensity due to ion suppression, however this can be offset by a better separation. As with most things a consideration has to be given to the both the separation and also the detection to ensure that the overall assay is optimised and focussing on one aspect will not always deliver success.
1. https://www.igeahub.com/2018/04/07/20-best-selling-drugs-2018/
