Columns (LC)
Impressive Separation of a Fusion Protein
Sep 05 2014
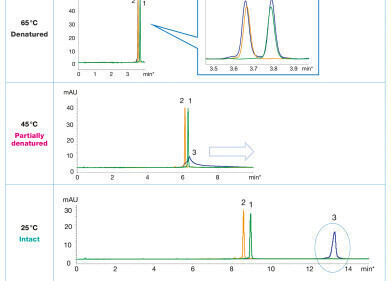
Thermo Fisher Scientific has developed a high-resolution hydrophobic interaction chromatography (HIC) method that can separate a fusion protein from a truncated version and a structural isomer. Application Brief 171: High-Resolution Separation of a Fusion Proteindemonstrates that two different HIC columns—the Thermo Scientific™ ProPac™ HIC-10 column and the new MAbPac™ HIC-10 column—can each perform this challenging separation. While the ProPac HIC-10 column is an established column for the analysis of monoclonal antibody post-translational modifications, the MAbPac™ HIC-10 column exhibits the best performance. The separation was also modified to facilitate fraction collection for further sample characterisation.
Fusion proteins are created through the genetic engineering of two or more genes that originated from separate proteins and impart properties from each of the parent proteins. Drugs based on fusion proteins have proven successful in clinical treatments. These protein therapeutics require high-resolution methods for characterisation and quality control techniques that are significantly different and more challenging than those used for small-molecule drugs. HIC can resolve proteins based on protein surface hydrophobicity, providing unique separation power complementary to other available techniques.
All trademarks are the property of their respective holders. Please note links are current at time of issuance.
Digital Edition
Chromatography Today - Buyers' Guide 2022
October 2023
In This Edition Modern & Practical Applications - Accelerating ADC Development with Mass Spectrometry - Implementing High-Resolution Ion Mobility into Peptide Mapping Workflows Chromatogr...
View all digital editions
Events
Apr 28 2024 Montreal, Quebec, Canada
May 05 2024 Seville, Spain
May 15 2024 Birmingham, UK
May 19 2024 Brno, Czech Republic
May 21 2024 Lagos, Nigeria