Columns (GC)
Published over 10 years ago. See the latest and most current information on Columns (GC).
Improved column inertness for polar analytes was the inspiration for the development of SLB-IL (i-series) ionic liquid capillary GC columns. They solve a dilemma that has existed for a long time; when analysing compounds with polar functionality, is it better to optimise for polar selectivity or polar inertness? With i-series columns, GC users can benefit from improved polar selectivity and polar inertness! Figure 1 illustrates the
resulting benefits.
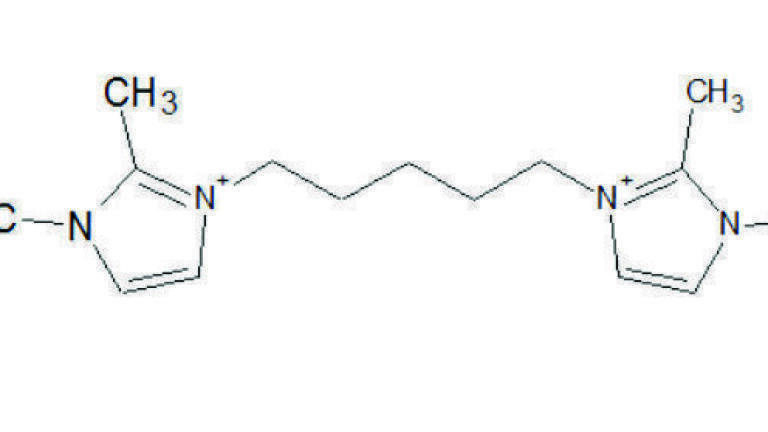
It is advantageous to have columns with alternative selectivity at hand, because resolution is most greatly affected by selectivity. A range of i-series columns were developed, classified as polar (SLB-IL60i), highly polar (SLB-IL76i), and extremely polar (SLB-IL111i). Table 1 contains complete specification for all three chemistries, whereas Figure 2 shows the structure of each stationary phase.
To demonstrate improved inertness, a polar column test mix containing four polar analytes and five n-alkane markers was analysed on two columns; SLB-IL76i (the improved inertness version) and SLB-IL76 (the original version). Both chromatograms are displayed in Figure 3. Using the polar column test mix for the quality control of polar and highly polar columns allows an assessment of inertness. In particular:
• The peak shapes of an alcohol, a ketone, a phenol (an aromatic alcohol), and an aniline (an aromatic amine) can be monitored – sharper peak shapes indicate a more inert column
• The % response of 1-octanol (its peak height relative to a curved line connecting the n-alkane markers) can be measured – a greater value indicates a more inert column
As shown, SLB-IL76i exhibits improved inertness, established by the sharp peak shapes produced for all analytes with polar functionality. This is further demonstrated by the greater peak height of 1-octanol.
Two by-products of the fermentation process are active amyl alcohol and isoamyl alcohol. Alcoholic beverage manufacturers are interested in these analytes due to their contribution to the aroma characteristic. A mix containing both analytes and several n-alkane markers was analysed on each i-series column. The chromatograms shown in Figure 4 reveal that all three chemistries are able to perform this separation using a 90°C isothermal oven temperature. Most other GC columns cannot perform this separation without resorting to lower (even sub-ambient) oven temperatures. Excellent peak shapes were exhibited for both analytes on all three chemistries. In fact, the alcohol peak shapes compare favourably to the n-alkane peak shapes! The inclusion of n-alkane markers allows some of the selectivity characteristic of each column to be revealed. These figures demonstrate the difference in selectivity between the three columns with the polar SLB-IL60i eluting the alcohols between n-undecane and n-dodecane. The more polar SLB-IL76i shifts the alcohols two carbon numbers downstream with them now eluting between n-tridecane and tetradecane. Finally, the extremely polar SLB-IL111i elutes the alcohols after the pentadecane peak. Having this range of selectivity and inertness will allow analysts the capability to tailor their analyses to shift components away from compounds that could interfere with their identification and quantitation.
Polar columns, such as those based on polyethylene glycol (PEG) stationary phase chemistry, are widely used for a variety of applications involving analytes with polar functionality, such as industrial solvents. However, modification of PEG phase chemistry to affect selectivity is limited. PEG type phases typically interact with compounds based on dispersive, hydrogen bonding and either acid or base type interactions. Columns based on ionic liquid stationary phases are able to undergo many of the same analyte-phase interactions as PEG columns, plus several additional interactions. The ionic liquid phases also have a dispersive, hydrogen bonding and acid base type interactions but in addition can also have possible pi-pi, dipole-dipole, dipole-induced dipole and possibly shape selective interactions. This results in unique selectivity which can be leveraged to change elution patterns and/or improve resolution. One example, though not shown is the separation of cis and trans fatty acid methyl esters (FAMEs). A PEG phase will elute the cis isomers as a group prior to the trans isomers. The similar polarity ionic liquid phase SLB-IL60i will elute the trans isomer group prior to the cis isomer group as is typically seen with highly polar cyanosilicone liquid phases like SP-2340.
An industrial solvent mix that contained 9 alcohols, 7 ketones, 10 esters, 3 ethers, 1 alkane, 12 aromatics, 11 chlorinated hydrocarbons, and 2 nitrogen-containing compounds was analysed on two columns using identical conditions. Figure 5 and Figure 6 show these chromatograms (SLB-IL60i and SLB-IL111i, respectively). Observations are that each column is able to produce sharp peak shapes for all analytes, and that complementary selectivity is achieved.
SLB-IL (i-series) columns offer improved inertness for applications involving complex mixtures comprised of compounds with varying functionality. Selectivity options exist, expanding their utility. Additionally, it is theorised that ideal GCxGC column sets can be assembled using these columns:
• SLB-IL60i paired with SLB-IL76i
• SLB-IL60i paired with SLB-IL111i
• SLB-IL76i paired with SLB-IL111i
Additional applications, product information, real-time availability, and ordering information is available at sigma-aldrich.com/il-gc-inert
