Columns (GC)
Published over 5 years ago. See the latest and most current information on Columns (GC).
Soft ionisation (chemical ionisation, field ionisation and photoionisation) methods are used in combination with comprehensive two-dimensional gas chromatography (GCxGC) coupled with high-resolution time-of-flight mass spectrometry (HRTOFMS). These techniques provide molecular weight and elemental composition information that is often absent from electron ionisation mass spectra and can simplify the extracted ion current (EIC) chromatograms in GCxGC-HRTOFMS analysis of complex mixtures. Examples are presented of field ionisation and photoionisation for biomarker identification in crude oil.
Comprehensive two-dimensional gas chromatography (GCxGC) [1-3] coupled with high-resolution time-of-flight mass spectrometry (HRTOFMS) is a powerful tool for the analysis of complex mixtures [4-7]. By using two columns with different stationary phases, GCxGC can achieve separations that cannot be attained with one-dimensional gas chromatography while HRTOFMS provides an additional dimension of separation by providing accurate masses for each mass-to-charge ratio.
Electron ionisation (EI) is widely used to produce mass spectra characterised by fragment ions that can be searched against mass spectral databases for compound identification. However, fragmentation adds an additional level of complexity in interpreting complex mixtures, such as crude oil. Furthermore, molecular ions can be weak or absent in the EI mass spectra of many compounds (e.g. alkanes, fatty acid esters, alcohols). Although the mass spectral databases contain entries for hundreds of thousands of compounds, these represent only a small fraction of all compounds that can be analysed by gas chromatography.
Soft ionisation methods suitable for GC-MS and GCxGC-MS include chemical ionisation (CI), photoionisation (PI) and field ionisation (FI). Each has its respective strengths and weaknesses.
Chemical ionisation is the most common soft ionisation method for GC-MS. This technique can be very sensitive for trace analysis and can be used for electron capture negative ion formation. However, CI requires reagent gases so ionisation, adduct formation and fragmentation are gas- and compound-dependent.
Field ionisation [8] [9] is the softest ionisation method for GCxGC-MS. For this technique a high voltage is applied to the emitter, which is a wire coated with tiny carbon ‘whiskers’. As the vaporised sample passes close to the tip of the ‘whiskers’ on the emitter, it experiences a high electric field, and electrons tunnel from the sample to the emitter, producing a positive ion. FI produces almost exclusively molecular ions for ‘difficult’ compound classed like alkanes and alcohols, and fragmentation is typically weak or absent. If a sample is deposited directly on the field emitter, direct probe analysis is possible. This type of experiment is referred to as field desorption (FD) [10] to distinguish it from FI, in which the sample is introduced in the gas phase. FI/FD emitters are fragile and expensive, but they can be reused hundreds of times. Also, FI is about an order of magnitude less sensitive than EI. Modern FI/FD sources on time-of-flight mass spectrometers are quite reliable and do not suffer from the high-voltage arcing problems that plagued FI/FD sources on earlier generation magnetic sector mass spectrometers. Jeol offers a combination EI/FI/FD source that permits switching between ionisation modes by using a probe and vacuum interlock to easily exchange the EI repeller and the FI or FD emitter.
Vacuum photoionisation [11-13] with a deuterium lamp ionises compounds with ionisation energies lower than the photon energy (10.8 eV). PI is simple to use with the Jeol combination EI/PI source by turning on the lamp and turning off the EI filament current. PI is more sensitive than FI for certain compound classes (e.g. PAH’s), but less sensitive for others (e.g. alkanes). It also produces slightly more fragmentation than FI but simpler spectra than CI.
These methods can be combined with high-resolution exact-mass data to simplify the processing of complex GCxGC data (e.g. petroleum type analysis), provide clear molecular weight and elemental composition that can be combined with EI data to identify unknowns, and provide selective ionisation of target compounds. Although GCxGC-HRTOFMS with soft ionisation is applicable to any GCxGC separation, it is particularly useful for petroleum analysis [6, 7, 14].
Petroleum biomarker analysis provides an example of the value of soft ionisation for GCxGC-HRTOFMS. Biomarkers are organic compounds that are present in petrochemicals and have characteristic distributions that reflect the original biological material from which the petrochemicals were formed [15]. Biomarkers measured by GC-MS and GCxGC-MS include steranes, phytane, and pristane. Biomarker analysis is used by petroleum geochemists to obtain information about the age, geographic origin and geographic distribution of oils. This information is important for petroleum exploration and forensic investigations of oil spills [16].
One-dimensional gas chromatography alone is insufficient to separate biomarkers in crude oil, so biomarker characterisation is commonly carried out by combined gas-chromatography and mass spectrometry. GC-MS methods rely on selected ion monitoring or better yet, high-resolution selected ion monitoring. Tandem mass spectrometry (GC-MS/MS) with selected reaction monitoring is an alternative approach that relies on the selectivity of MS/MS for high sensitivity measurements. On the other hand, comprehensive two-dimensional gas chromatography combined with high-resolution time-of-flight mass spectrometry provide greater separation, better signal-to-noise, and greater selectivity for isomeric biomarkers [17]. Additionally, FI and PI can be applied to biomarker analysis with GCxGC-HRTOFMS to simplify data interpretation by reducing fragmentation [5]. In this work, petroleum biomarkers were identified in crude oil by using FI and PI with GCxGC-HRTOFMS.
An Agilent 7890B gas chromatograph fitted with a Zoex ZX-2 GCxGC thermal modulator was interfaced to a Jeol JMS-T200GC time-of-flight mass spectrometer equipped with a combination EI/PI ion source and EI/FI/FD ion source. A nonpolar BPX5 column (SGE Corporation, 30 m length, 0.25 mm I. D., 0.25 μm liquid phase thickness) was used for the first column. A more polar BPX50 column (SGE Corporation, 3 m length, 0.1 mm I. D., 0.1 μm liquid phase thickness) was used for the second column.
Experimental conditions are given in Table 1.
Figure 1 shows the EI mass spectra for 5-a-cholestane (C27H48) and 5-a-stigmastane (C29H52) from the NIST 2020 Mass Spectral Database and the measured photoionisation
and field ionisation mass spectra for these compounds. Although the EI mass spectra exhibit molecular ions, extensive fragmentation is also observed, including methyl loss to produce fragment ions 12C27H37+ and the isotope 13C112C26H37+.
Both the PI and FI mass spectra show abundant molecular ions with drastically reduced fragmentation. Little or no fragmentation is observed for the FI mass spectra, but the PI mass spectra show a characteristic C16H26 sterane fragment[16] at m/z 218.204.
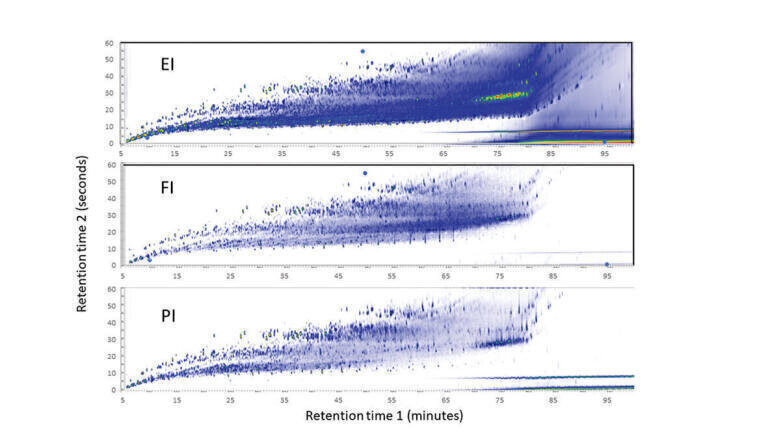
The 2D total-ion chromatograms for GCxGC-HRTOFMS of the standard crude oil sample using EI, FI and PI are shown in Figure 2. The x-axis represents separation by boiling point on column 1 and the y-axis represents separation by polarity on column 2. Although the EI chromatogram shows higher background and column bleed than the FI and PI chromatograms, the chromatograms are otherwise identical.
To identify cholestane isomers (C27H48) in the crude oil, we created an extracted ion current (EIC) chromatogram (referred to as ‘Selected Ion Chromatogram’ in the GC Image software) for the exact mass for the molecular ion (m/z 372.3756). However, fragmentation in the EI mass spectra is problematic [18] because of interference from the heavy isotope peaks for the fragment ions from higher cholestane homologues. For example, loss of a methyl group from 14-methyl cholestane (C28H50) produces a fragment ion in the EI mass spectrum with the composition C27H47. The 13C isotope peak for this fragment is found at m/z 372.3712, which requires a resolving power of over 200,000 to distinguish it from the cholestane molecular ion C27H48+•. However, this resolving power is not available for mass spectrometers that can be operated at the high speeds required for GCxGC.
As shown in Figure 1, field ionisation and photoionisation of cholestanes do not exhibit fragmentation to C27H47+. Therefore, the FI and PI EIC chromatograms for m/z 372.3756 do not exhibit any interference from isotope peaks of fragment ions of higher homologues. Figure 3 compares the electron ionisation EIC chromatogram for m/z 372.3756 with the FI and PI EIC chromatograms for the same m/z. Although the EI data show artifact peaks from 13C112C26H37+, these artifacts are absent in the FI and PI data. Twenty isomers of cholestane can be clearly identified in the artifact-free FI and PI data (Figure 4).
Comprehensive two-dimensional gas chromatography combined with high-resolution time-of-flight mass spectrometry provides enhanced separation and specificity for the analysis of complex mixtures such as petrochemicals. Although electron ionisation produces mass spectra that can be searched against the mass spectral databases, the extensive fragmentation in EI mass spectra complicates the data interpretation. By reducing or eliminating fragmentation artifacts, field ionisation and photoionisation provide greater specificity for petroleum biomarker and type analysis. These ion sources are available with the newest Jeol time-of-flight mass analyser (JMS-T2000GC) with greatly enhanced mass resolving power and mass accuracy that is expected to provide even greater accuracy and specificity for these analyses.
1. Z. Liu, J. B. Phillips. Comprehensive Two-Dimensional Gas Chromatography using an On-Column Thermal Modulator Interface. Journal of Chromatographic Science, 1991, 29, 227.
2. R. C. Y. Ong, P. J. Marriott. A Review of Basic Concepts in Comprehensive Two-Dimensional Gas Chromatography. Journal of Chromatographic Science, 2002, 40, 276.
3. H. J. Cortes, B. Winniford, J. Luong, M. Pursch. Comprehensive two dimensional gas chromatography review. Journal of Separation Science, 2009, 32, 883.
4. M. Ubukata, K. J. Jobst, E. J. Reiner, S. E. Reichenbach, Q. Tao, J. Hang, Z. Wu, A. J. Dane, R. B. Cody. Non-targeted analysis of electronics waste by comprehensive two-dimensional
gas chromatography combined with high-resolution mass spectrometry: Using accurate mass information and mass defect analysis to explore the data. Journal of Chromatography A, 2015, 1395, 152.
5. M. Ubukata, Y. Ueda. Biomarker Analysis in Petroleum Samples Using GC×GC-HRTOFMS with an Ion Source Combining Electron Ionization (EI) and Photo Ionization (PI). JEOL News, 2018, 53, 54.
6. A. Giri, M. Coutriade, A. Racaud, K. Okuda, J. Dane, R. B. Cody, J.-F. Focant. Molecular Characterization of Volatiles and Petrochemical Base Oils by Photo-Ionization GC×GC-TOF-MS. Analytical Chemistry, 2017.
7. A. Giri, M. Coutriade, A. Racaud, P.-H. Stefanuto, K. Okuda, J. Dane, R. B. Cody, J.-F. Focant. Compositional elucidation of heavy petroleum base oil by GC × GC-EI/PI/CI/FI-TOFMS. Journal of Mass Spectrometry, 2019, 54, 148.
8. H.-D. Beckey, Field Ionization Mass Spectrometry, Pergamon (Elsevier), Oxford, 1971.
9. N. M. M. Nibbering, in The Encyclopedia of Mass Spectrometry (Eds.: M. L. Gross, R. M. Caprioli), Elsevier, Boston, 2016, pp. 92.
10. L. Prókai, Field Desorption Mass Spectrometry, Marcel Dekker, Inc., New York and Basel, 1989.
11. F. P. Lossing, I. Tanaka. Photoionization as a Source of Ions for Mass Spectrometry. J. Chem. Phys., 1956, 25, 1031.
12. J. C. Traeger, in Encyclopedia of Spectroscopy and Spectrometry (Third Edition) (Eds.: J. C. Lindon, G. E. Tranter, D. W. Koppenaal), Academic Press, Oxford, 2017, pp. 650.
13. D. J. Butcher. Vacuum Ultraviolet Radiation for Single-Photoionization Mass Spectrometry: A Review. Microchemical Journal, 1999, 62, 354.
14. K. Qian, F. C. Wang. Compositional Analysis of Heavy Petroleum Distillates by Comprehensive Two-dimensional Gas Chromatography, Field Ionization and High-resolution Mass Spectrometry. Journal of The American Society for Mass Spectrometry, 2019, 30, 2785.
15. C. S. Hsu, C. C. Walters, G. H. Isaksen, M. E. Schaps, K. E. Peters, in Analytical Advances for Hydrocarbon Research (Ed.: C. S. Hsu), Springer US, Boston, MA, 2003, pp. 223.
16. Z. Wang, S. A. Stout, M. Fingas. Forensic Fingerprinting of Biomarkers for Oil Spill Characterization and Source Identification. Environmental Forensics, 2006, 7, 105.
17. N. G. S. Mogollón, P. S. Prata, F. Augusto.
Determination of Biomarkers in Petroleum by Multidimensional Gas Chromatography: Fundamentals, Applications, and Future Perspectives. Chromatography Today, 2014, May/June, 3.
18. K. E. Peters, C. C. Walters, J. M. Moldowan, The Biomarker Guide: Volume 1: Biomarkers and Isotopes in the Environment and Human History, Vol. 1, 2 ed., Cambridge University Press, Cambridge, 2004.
