-

-
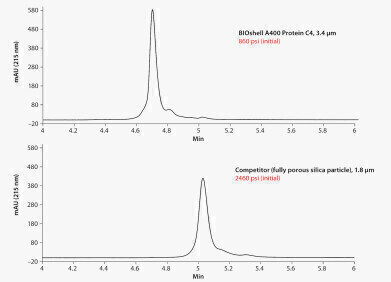 Figure 1.
Figure 1. -
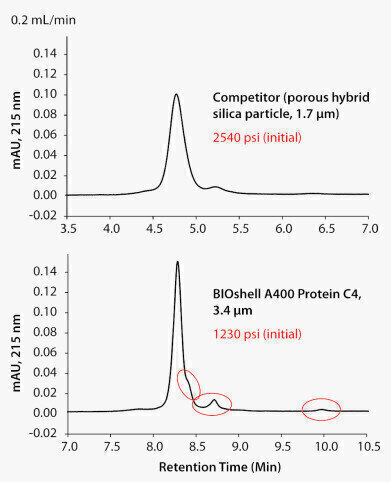 Figure 2.
Figure 2.
HPLC, UHPLC
Optimised HPLC Analysis of Monoclonal Antibodies (mAbs)
Nov 23 2016
Monoclonal antibodies (mAbs) are complex macromolecules that are developed as biotherapeutics themselves, or as targeting vehicles for delivery of toxic payloads (antibody drug conjugates). As complex macromolecules, they require multipronged approaches to analysis and characterisation, whether under development or to support production QC. Analysis of the intact mAb, can provide first level assessment of size, post-translational modifications, and heterogeneity. Such questions can be readily addressed by reversed-phase chromatography (RPC) coupled to the analytical power of mass spectrometry. RPC of proteins, particularly as large as IgG, requires the use of a large pore chromatographic matrix, a hydrophobic stationary phase, and appropriate chromatographic conditions – mobile phase and temperature. The typical phase for RP protein chromatography is a short alkyl chain, which provides adequate retention. By way of RP mobile phase conditions, however, mAbs generally require an elevated temperature for efficient chromatography. It has also been reported that the chromatography can be further enhanced by the inclusion of a protic solvent as a component of the organic modifier (the strong solvent). In addition to optimising the chromatographic conditions, new developments in particle technology have resulted in gains in chromatographic performance. These technology developments have been applied to both traditional porous particles and new core-shell (or superficially porous) particle design. For porous particles, the advancements have come in smaller particle size and chemical stability. For core-shell particles, the technological advancement is in the particle architecture itself - limiting the chromatographic separation process to the thin outer porous layer. Additionally, the synthetic route to the core-shell particle may yield a narrower particle size distribution, which in turn can affect chromatographic packing such that improved column efficiency is attained.
The figures compare the chromatographic results of two such modern columns designed for intact protein analysis. In Figure 1, the sample is the mAb adalimumab (aka Humira®), while for Figure 2, the sample is a mAb standard from MilliporeSigma (MSQC4). Both new columns were run on the same day with the same sample, on the same UHPLC instrument. A common method was used for both columns, at a temperature optimum of 75°C. It is clear that the core-shell column provides considerably narrower peak width (and thus better sensitivity), and as a result of the superior performance, also better resolves minor components of the sample. The enhanced performance of the core-shell column will be best realised in combination with a modern UHPLC instrument of minimal system dispersion. Please click here for additional information.
Chromatographic Conditions:
| column | as indicated, 10 cm x 2.1 mm I.D. |
| mobile phase A | 80:20, (water, 0.1% TFA):(acetonitrile, 0.1% TFA) |
| mobile phase B | 50:50, (water, 0.1% TFA):(acetonitrile, 0.1% TFA) |
| flow rate | 0.2 mL/min |
| gradient | 45 to 57% B in 12 min |
| column temp | 75°C |
| det | UV, 215 nm |
| injection | 0.3 µL Humira or 1.0 µL SigmaMAb |
| sample | mAb diluted in 20 mM Tricine/KOH, pH 7, to 1 g/L |
Digital Edition
Chromatography Today - Buyers' Guide 2022
October 2023
In This Edition Modern & Practical Applications - Accelerating ADC Development with Mass Spectrometry - Implementing High-Resolution Ion Mobility into Peptide Mapping Workflows Chromatogr...
View all digital editions
Events
ACS National Meeting - Fall 2024
Aug 18 2024 Denver, CO, USA
Sep 04 2024 Chiba, Tokyo, Japan
Sep 04 2024 University of Warwick, Coventry, UK
Sep 10 2024 Rockville, MD, USA
Plastics Recycling World Expo Europe
Sep 11 2024 Brussels, Belgium













