-
 Continuous validation of HPLC performance.
Continuous validation of HPLC performance. -
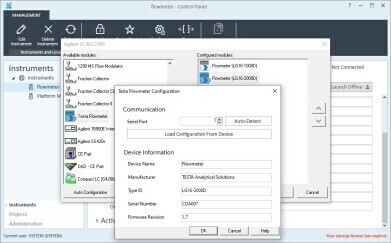 Flowmeter Software Driver for Chromatography Data Systems. (courtesy: Testa Analytical)
Flowmeter Software Driver for Chromatography Data Systems. (courtesy: Testa Analytical)
Data Handling
Reliable HPLC performance validation in a regulated lab
Dec 18 2023
Testa Analytical has reported the successful testing of its flowmeter software driver in a regulated pharmaceutical laboratory setting. With a focus on seamless integration with various HPLC and GPC/SEC manufacturers' chromatography software packages, the software underwent thorough testing during its development cycle.
Jeanette Ziemba, Technical Manager at Testa Analytical, highlighted the software's extensive evaluation in a leading European pharmaceutical lab. The lab, managing diverse HPLC instruments with different chromatography data systems in a tightly regulated client-server environment, praised the software for its ability to facilitate easy acquisition and traceable logging of flow data from all liquid chromatographs. The software's support for multiple Flowmeters connected to different HPLC systems in both analytical and semi-prep applications was particularly appreciated for its simplifying effect.
Ziemba emphasised the value of saving real-time flow rate data with each chromatogram, citing it as a crucial aid in achieving total quality assessment for various chromatographic systems. The software driver, built on rc.NET technology, demonstrated cross-platform connectivity with Testa Analytical flowmeters and popular Chromatography Data System software packages, including OpenLab (Agilent Corp), Clarity Chromatography Software (DataApex), Empower (Waters Corp), LabSolutions (Shimadzu Corp), and WinGPC Software (Agilent).
She concluded by underlining the software's ability to collect and store real-time flow rate data, along with chromatography detector signals, as a means of enhancing quality assessment and providing continuous validation of compliance with the planned chromatographic separation method. The affordable flowmeter software driver is positioned to complement and extend the capabilities of modern liquid chromatography software packages.
More information online
Digital Edition
Chromatography Today - Buyers' Guide 2022
October 2023
In This Edition Modern & Practical Applications - Accelerating ADC Development with Mass Spectrometry - Implementing High-Resolution Ion Mobility into Peptide Mapping Workflows Chromatogr...
View all digital editions
Events
ACS National Meeting - Fall 2024
Aug 18 2024 Denver, CO, USA
Sep 04 2024 Chiba, Tokyo, Japan
Sep 04 2024 University of Warwick, Coventry, UK
Sep 10 2024 Rockville, MD, USA
Plastics Recycling World Expo Europe
Sep 11 2024 Brussels, Belgium













