GC-MS
Published over 6 years ago. See the latest and most current information on GC-MS.
The analysis and detection of performance-enhancing drugs in sports competitions can be challenging due to the continuous development of new drugs and the continuous abuse of established and discontinued drugs. This article highlights a complete workflow solution including sample preparation for different doping substances, as well as confirmation and quantification using GC-QqQ and GC-HRAM.
Gas chromatography combined with mass spectrometry (GC-MS) is widely used for the analysis of sports doping samples [1]. However, these workflows are often highly complex, involving multiple steps from the collection of samples through to the reporting of results. In animal doping analysis especially, the sample matrix can pose a challenge in terms of sample preparation. Horse urine, for example, is often sent to the testing laboratory in gel form since horse urine forms a gel, when exposed to air, and extensive sample preparation is required before analysis can take place. Following sample preparation, other considerations such as the use of appropriate glass vials and inert glass liners in the GC instrument, both should be made from borosilicate type I glass [2], must be taken into account to ensure analytes do not adsorb to the free silanol groups on the surface and impact on the accuracy and reliability of results.
Triple quadrupole instruments (QqQ) are traditionally used for MS detection in the analysis of sports doping samples. However, advances in high resolution-accurate mass technology (HRAM) mean that this approach offers several benefits over QqQ detection methods. This article highlights a complete workflow solution for the analysis of a broad range of drugs in urine using GC-QqQ and GC-HRAM methods. An assessment of detection limits and system robustness for a workflow used by the Rio de Janeiro Doping Control Laboratory (LBCD–LADATEC) during the 2016 Summer Olympic Games is presented.
Sample preparation and derivatisation
Liquid-liquid extraction (LLE) is widely used for the preparation of urine samples prior to analysis. However, this approach is non-selective and can result in the extraction of other matrix materials, such as urea, hormones and other substances that urine might contain. Furthermore, LLE is not always suitable for the extraction of all analytes and does not involve a concentration step, which can impact measurement sensitivity. In SPE you start with 3 ml sample and end up with 50 µl of sample prior to injection after drying down.
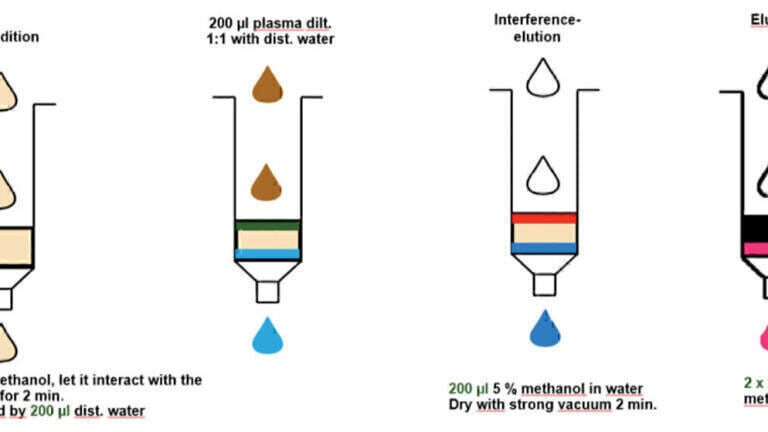
In the workflow reported here, a sample preparation step utilising either a C18 or cation exchange solid phase extraction (SPE) cartridge was used, depending on the substance class to be analysed. To provide the cleanest results possible, silica-based materials were employed, as polymers can release substances that can interfere with the measurement and lead to higher limits of detection (LODs) and limits of quantitation (LOQs) [3].
Figure 1 and Figure 2 highlight the SPE protocols using the C18 and mixed-mode cation exchanger, respectively. Silica-based C18 and mixed-mode cation exchangers were used as these resulted in the cleanest extracts for the GC-MS analysis. The figures show generic protocols, which were employed for the majority of the analytes studied. However, modifications to the method were required in some cases. Additionally, for the horse urine samples, it was necessary to freeze the samples in liquid nitrogen prior to the SPE step. Freezing opens up the cells and gets the urine back in a liquid form. Adding other chemicals to get the urine in a liquid form can reduce the activity of the beta-Glucuronidase.
SPE yields cleaner extracts and can offer a concentration factor of up to 60-fold, resulting in enhanced LODs. As explained above, the starting point is 3000 µl urine and the final sample volume is 50 µl prior to injection. LLE does not provide concentration enhancement, and the extracts are not clean, which can lead to higher detection limits when analysed by GC-MS.
After SPE, the samples were evaporated and reconstituted and/or derivatised. Silylation was used for those substances that contained, COOH-, NH- or NH2 groups in order to reduce the polarity of the substance. Underivatised they would possibly stick in the glass vial or liner. The derivatisation step was fully automated using a Thermo Scientific Triplus RSH Autosampler. This enabled batch derivatisation in dedicated preparation cycles (a batch of 6 samples was derivatised prior to injection) or the use of a sequential procedure in which each sample was derivatised and injected. Prior to injection, the samples were transferred into Thermo Scientific Chromacol GOLD-Grade inert glass vials (Type 1 hydrolytic class glass, 29% free silanol groups on the surface) to minimise adsorption to the vial surface. [4]
Following sample preparation, the samples were injected into the GC-MS system. The samples were separated using a Thermo Scientific TRACE 1310 GC system coupled with a Thermo Scientific TSQ 8000 triple quadrupole mass spectrometer. The triple quadrupole MS data were compared to an Q Exactive Orbitrap mass analyser to see, what benefits in resolution Orbitrap can provide. Table 1 shows the GC and GC-MS parameters used.
GC-QqQ offers good selectivity for endogenous and exogenous steroids, as well as other relevant analytes of interest in urine. This technology is widely used and is well established in antidoping laboratories around the world. For the non-targeted approach, results are shown on a GC-HRAM with 1 ppm mass accuracy across a broad linearity range, including good sensitivity, and linearity for a large number of compounds.
GC-QqQ enables doping control laboratories to perform screening, confirmation, and quantification of analytes in complex matrices in a single analysis without the need to change their current extraction and GC separation methods. The identification and quantitation of anabolic-androgenic steroids (AAS) is well studied using GC-QqQ technology. GC-QqQ offers significant benefits over LC-MS interfaced to atmospheric ionisation sources, which are limited by poor ionisation. Selected reaction monitoring (SRM) provides trace-level detection of the steroids of interest, separating the chemical noise of the matrix from the signal and assuring high sensitivity and selectivity even in very complex matrices, such as camel and horse urine.
GC coupled with single quadrupole MS in selected ion monitoring (SIM) mode has been widely used for anti-doping applications, and it is still used in laboratories for many applications. However, for steroids analysis, single quadrupole systems are limited in terms of selectivity and sensitivity [1].
Figure 3 shows the selectivity of single versus triple quadrupole systems for a real urine sample containing fluoxymesterone-M2. The results highlight that the single quadrupole system did not provide the necessary sensitivity and selectivity. Co-elution’s from the matrix make it difficult to determine the identity of the analyte and also reduce the sensitivity of the measurement. SRM using the triple quadrupole system, on the other hand, provides both excellent sensitivity and selectivity.
The Rio de Janeiro Doping Control Laboratory (LBCD–LADATEC) analysed approximately 500 samples per day during the 2016 Summer Olympic Games. To meet these demanding sample throughput requirements, robust and reliable analytical workflows were essential. Table 2 shows the World Anti-Doping Agency Minimum Required Performance Levels (WADA/MRPL) and the LOD of banned substances and their metabolites.
Table 2. Limits of detection.
Bromantane-6OH and Fluoxymesterone M2 (17,17-dimethyl-18-nor) were not analysed in this experiment, Testosterone-D3 is used as an internal standard.
GC-HRAM enables a simultaneous quantitative and qualitative analysis of exogenous and endogenous steroids. Orbitrap MS provides excellent sensitivity and high mass accuracy in the SRM workflow, and exceptional linear range is demonstrated, especially for endogenous steroids. Spectral fidelity is maintained across the required concentration range including mass accuracy. Additional benefits of the non-targeted acquisition workflow include the ability to perform retrospective data analysis and post-acquisition data processing, enabling future investigations to be made on existing samples, should new compounds of interest be identified.
The electron energy also plays an important role in the determination of steroids. Reducing the energy from 50 to 30 eV resulted in a 2.5–10-fold decrease in LODs. Reducing the electron energy further did not lead to additional sensitivity improvements. The GC Orbitrap delivered optimal results for the steroids at an electron energy of 30 eV. Table 3 shows the linearity and LOD for the exogenous steroids, while Figure 5 highlights the benefits of using an exact mass GC for endogenous steroids.
The high mass accuracy allows lower detection limits utilising the 5th decimal after the comma in the exact mass, to detect apexing coeluting substances and to get rid of disrupting background m/z. This is not possible using QqQ, since those are nominal m/z instruments.
This study highlights the exceptional results generated from this complete GC-MS workflow. Several factors were important in delivering these results, including the choice of sample preparation method and glassware [4].
The GC-QqQ system showed the expected results in an anti-doping laboratory under routine conditions for screening, confirmation and quantification in a single analysis workflow. The SRM approach delivered trace-level detection of the analytes of interest as required by WADA/MRPL methods.
Using HRAM Orbitrap technology-enabled simultaneous qualitative and quantitative analysis of exogenous and endogenous steroids. The high selectivity of the exact mass measurements could also be used to determine new substances. Additionally, the wide linear range and the potential for non-targeted analysis makes GC-HRAM a valuable complementary method to GC-QqQ for anti-doping applications.
We would like to thank Sporthochschchule Köln, Landschaftsverband Rheinland and UFRJ Instituto de Chimica for their support with this study.
1. ICRAV 2018, Dubai UAE, Friday 9th of March, Workflow solution for Steroids and Anti-Doping Analysis in Urine with GC-MS/MS and GC HRAM
2. European Pharmacopoeia 7, 01/2008, Chapter 3.2., Glass Containers for Pharmaceutical Use
3. Thermo Fisher Scientific Webinar, 25.7.2019, Fundamentals of Solid Phase Extraction
4. White Paper 21833, Thermo Fisher Scientific, Lennartz et al,
Why is high glass quality so important when you want to detect low concentrations of analytes?
