Chiral
Published over 9 years ago. See the latest and most current information on Chiral.
Ultra High Performance Chromatography (UHPC) Whelk-O1 1.8 µm Chiral Stationary Phase (CSP) was evaluated from the kinetic point of view through van Deemter curves analysis on trans-stilbene oxide under Normal Phase conditions and compared to commercially available columns. The sub-2µm CSP exhibited more than 260,000 theoretical plates/m on the first eluted enantiomer at an optimal flow-rate of 1.8 mL/min (T: 35°C), obtaining efficiencies more than three times higher than the Regis HPLC- Whelk-O1 5 µm and, at the same time, maintained very high efficiencies at large linear velocities. High reproducibility in terms of thermodynamic performances (retention factor (k’) and selectivity (a)) was recorded between 1.8 µm CSP and the 3.5 and 5 µm columns. This chiral stationary phase was also employed in sub-critical fluid conditions. A van Deemter curves analysis was performed maintaining efficiencies of more than 250,000 theoretical plates per meter. This chiral selector was also used in the Ultra High Performance Super Criticalfluid Chromatography (UHPSFC) screening, in a single day, of a large library of 129 acidic, neutral and basic racemates employing a 9-min screening run using a 5 cm long column.
Over the last ten years, technological advances have led to the development of achiral stationary phases bonded on ever smaller silica particles (sub-2µm) and instrumentation (UHPLC/UHPSFC) with a reduced extra-column volume able to reach very high pressures. These improvements allow the user to obtain high efficiencies and resolutions, and to reduce analysis time and eluent consumption. However, only in the last few years have chiral stationary phases started moving in this direction [1-2]. In enantio-chromatography, research has been focusing on the design and development of selectors with a very broad spectrum, with the aim to resolve the highest number of racemates. Since there is not yet a unique and universal Chiral Stationary Phase (CSP), many CSPs are needed [3-17]. Recently many CSPs have been covalently bonded on very small silica particles, Fully Porous Particles (FPP) and Superficially Porous Particles (SPP). The 3,5-dinitro- benzoyl derivative of 1,2- diaminocyclohexane (DACH-DNB) [18], β-cyclodextrin [19], Whelk-O1 [2-20-21], cyclofructan derivatives [22] and macrocyclic antibiotics [8,23] are all examples of covalently bonded phases
A new sub-2µm Ultra High Performance Chromatography (UHPC) chiral stationary phase was developed using Whelk-O1 as a selector, bonded on 1.8 µm Kromasil silica, with the aim to obtain very high efficiencies, very high resolutions and fast separations [24]. Whelk-O1 is a Pirkle type selector, introduced firstly by Pirkle in 1992 [11,25], with a broad application field. Aryl propionic NSAIDs and all compounds that have aromatic systems with hydrogen bond acceptor sites near the chiral centre [26] are ideal targets for the Whelk-O1 CSP. Because of the fast interaction provided by the Whelk-O1 selector efficiencies of more than 260,000 theoretical plates/m were achieved on trans-stilbene oxide in Normal Phase – Ultra High Performance Liquid Chromatography (NP-UHPLC) analysis. UHPSFC is also a valid alternative analysis technique [27]. In fact, because of the low viscosity of the CO2 high flow-rates with low backpressure can be achieved. SFC is an excellent choice to perform very fast analysis with high resolution values, allowing high throughput enantioselective screening [21] and reducing the consumption of organic solvents.
The UHPLC system used for all tests was an UltiMate 3000 RS system from Thermo Fisher Dionex (Sunnyvale, California), consisting of a dual gradient RS pump (800 bar under normal phase conditions; flow rates up to 8.0 mL/min), an in-line split loop Well Plate Sampler, a thermostated RS Column Ventilated Compartment (temperature range 5-110°C) and a diode array detector (UV Vanquish detector) with a low dispersion 2.5 μL flow cell. The UV Vanquish detector was set at a filter time constant of 0.002 s, a data collection rate of 100 Hz and a response time of 0.04 s. Viper capillaries and fittings were used, with the two capillary Viper tubes (2 x 350 mm × 0.10 mm I.D.). Data acquisition and processing was performed with Chromeleon 6.8 software from Thermo Fisher. Detection of tested analytes was carried out at two different wavelengths (214 nm and 220 nm). The extra-column volume (obtained by injecting naphtalene) of this equipment was 12.7 μL (variance,s2v,extra= 6.86 μL2 at flow-rate 1.0 mL/min, calculated with second statistical moment, eluent: Hexane/EtOH 90:10 + 1% MeOH, T: 35°C) [28].
A Waters Acquity UPC2 (Ultra Performance Convergence Chromatography) was used to perform SFC analyses. The system was equipped with a binary solvent delivery pump compatible with mobile phase flow rates up to 4 mL/min and maximum system pressure of 414 bar. A 250 mL mixing chamber is present in the delivery system. The system also comprised an autosampler with a 10 mL loop, a column oven compatible with temperatures up to 90°C, an UV detector equipped with an 8 mL flow–cell and a backpressure regulator (BPR). The injector/column inlet and column/detector connection tubes were 600 mm long and had an I.D. of 0.175 mm. The extra-column volume of this instrument was estimated to be 60 mL [29]. Data acquisition and control of the UHPSFC system was performed with the Empower 3.
Solvents and reagents were from Sigma-Aldrich (St. Louis, Mo, USA) and used without further purification. HPLC grade solvents were filtered on 0.2 mm Omnipore filters (Merck Millipore, Darmstadt, Germany). Kromasil silica from Akzo Nobel (pore size 100 Å, particle size 1.8 mm and specific surface area 320 m2 g−1), Whelk-O1 selector and the two commercially available columns Whelk-O1 150x4.6 mm 3.5 and 5 mm were a gift from Regis Technologies Inc®. 10 cm and 5 cm long empty stainless steel columns with an internal diameter of 4.6 mm were from IsoBar Systems by Idex (Wertheim-Mondfeld, Germany).
Synthesis of UHPLC Whelk-O1 CSP
The UHPC-Whelk-O1 chiral stationary phases (CSPs) were synthesised according to the procedure described by Pirkle in 1992 [11,25]. All UHPLC-Whelk-O1 columns were slurry packed with a pneumatically driven Haskel pump (Pmax: 1000 bar) into 100 x 4.6, 50 x 4.6 mm L.xI.D.
The kinetic performances of this new sub-2µm CSP were evaluated under Normal Phase conditions (NP) and Super/sub-critical fluid chromatography (SFC). The mobile phases used for all tests were a mixture of Hexane/Ethanol 90:10 + 1% MeOH and CO2/MeOH 80:20 in NP and SFC conditions respectively. All injections were performed setting a Vinj of 0.1-0.5 mL in isocratic elution mode, repeated twice and the average values were used in the van Deemter construction. Kinetic evaluation were performed by the analysis of van Deemter curves starting from a minimum flow-rate of 0.1 mL/min up to 3.0 mL/min in NP and up to 4.0 mL/min in UHPSFC at the temperature of 35°C. All data were processed with Origin 6.0 in order to properly graph and fit the van Deemter curves. Efficiencies (N/m or plates/m), and height theoretical plates (H), were not corrected for extra-column band broadening. Void volume was obtained by injecting an unretained marker (CCl4 in NP and CHCl3 in UHPSFC).
The resolution (Rs), efficiencies and peak asymmetry values were extracted from Chromeleon 6.8 or Empower 3.0 softwares (according to European Pharmacopeia using peak width at half height (W0.5)).
Kinetic performances of the new UHPC-Whelk-O1-1.8 columns (100 x 4.6 mm) were evaluated under UHPLC conditions through van Deemter curves analysis, correlating the efficiency, expressed as theoretical plate height, H (µm), with the flow-rate (mL/min) at the temperature of 35°C. Plots were compared to those of the commercially available Whelk-O1 3.5 µm and 5 µm 150x4.6 mm (Figure 1A). Looking at the UHPC-Whelk-O1-1.8 curves, there is a clear advantage obtained in terms of efficiency. Efficiencies higher than 260,000 plates/meter were recorded on the first eluted enantiomer of trans-stilbene oxide versus the almost 140,000 plates/meter on the 3.5µm and the only 70,000 of the 5 µm. The same trend was observed for the second eluted enantiomer with a gain in efficiency of 75% going from the commercial 3.5 µm to UHPC-Whelk-O1-1.8. Taking into consideration the 5 µm Whelk-O1 in comparison with the sub-2µm CSP, the efficiency was increased more than 3.5 times (68,000 versus more than 250,000 N/m) A large gain in optimal flow-rate range was also observed. Maximum efficiencies were recorded at 1.8, 1.1 and 0.6 mL/min, for the first enantiomer, on the 1.8 µm, 3.5 µm and 5 µm respectively. This permits the use of higher flow-rates with only a small loss of efficiency at the optimal flow-rate (more than 63% efficiency loss on the Whelk-O1 5 µm versus only 13% efficiency loss on the 1.8 µm for the first enantiomer at flow-rate: 3.0 mL/min). This trend can clearly be observed in Figure 1B, where the column efficiency was correlated to the flow-rate. Looking at this graph it is easy to understand the kinetic improvement of the 1.8 µm that yielded higher optimal flow-rates and better efficiencies with a 10 cm long column compared to 3.5 and the 5 µm commercial CSP packed into 15 cm columns. In Figure 2A are shown the chromatograms obtained injecting trans-stilbene oxide on the three columns at the respective optimal flow-rates. The separation provided by the UHPC-Whelk-O1 1.8 µm (100 x 4.6 mm ) was completed in just 1.65 minutes, with a gain of more than four times in speed compared to the 5 µm commercial column (150 x 4.6 mm). One of the most important advantages, due to the kinetic profile described previously, is the resolution (Rs), which was 17.74 on the 100x4.6 mm 1.8 µm Whelk-O1, almost two times larger than the 150x4.6 mm 5 µm column. The thermodynamic profile was characterised in Figure 2B, where the same chromatographic traces of Figure 2A were reported with the k’ on the x-axis, in order to evaluate the performances of these columns independently of the column geometries. Extremely reproducibility k’, and selectivity, was achieved between the 1.8 µm Whelk-O1 and the two commercially available Whelk-O1 columns. In fact, an enantioselectivity factor of 2.42-2.49 was recorded on all columns. This reproducibility is extremely important in the transition from enantioselective ‘e’ High Performance Liquid Chromatography (eHPLC) to ‘e’ Ultra High Performance Liquid Chromatography (eUHPLC). Using 1.8 µm columns instead of the 3.5 µm (or 5 µm) without any change in the analytical methodology, maintaining the same conditions (i.e. mobile phase composition), it is possible to improve the separation of two enantiomers with better efficiencies and resolutions thus improving the speed of analysis and reducing the analysis time.
Figure 3 shows two different separations, 1-acenapthenol (Figure 3A) and benzoin (Figure 3B), on the UHPC-Whelk-O1 1.8 µm 100x4.6 mm and on the two Regis Whelko-O1 150x4.6 mm 3.5 and 5 µm. In both cases the sub-2µm, Whelk-O1 reached efficiencies two and four times greater in comparison with the 3.5 and the 5µm HPLC Whelk-O1 CSPs. All injections were performed at the optimal flow-rate of each column, increasing the speed of analysis by 5 times and enhancing the resolution values more than 30%. With the very high reproducibility obtained on the UHPC-Whelk-O1, the retention factors and the enantioselectivity values were almost the same, when using the same mobile phase, on all columns and on both samples.
This phase was also used in Sub/super-critical fluid chromatography with a mobile phase consisting of 80% CO2 and 20% methanol. van Deemter curves analysis was performed also in this case (Figure 4). Efficiencies up to 250,000 were recorded on the sub-2µm Whelk-O1 with a gain of more than three times in comparison to the 5µm column. Also the optimal flow-rate was more than twice that of the commercial Whelk-O1. Another interesting aspect is the optimal linear velocity, which can’t be achieved with the current instrumentation (UPC2 has an instrumental limit at 4 mL/min and 6000 psi). The curves of the two enantiomers are not able to reach the minima, and those can only be estimated at an optimal flow-rate of 5-6 mL/min [27] and consequently it was not possible to record maxima efficiencies of the column.
Figure 5 shows the chromatographic traces recorded on trans-stilbene oxide on the three columns in SFC conditions at their optimal flow-rates. Under these conditions, an analysis time gain of more than three times was observed and the separation of the two enantiomers was completed in only 1.08 minutes at the flow-rate of 3.7 mL/min (instrumental pressure limit) in comparison to the 5 µm. Also the resolution was extremely improved going from 13.5 to 19 and up to 21.5 on the 5, 3.5 and 1.8 µm Whelk-O1 columns respectively.
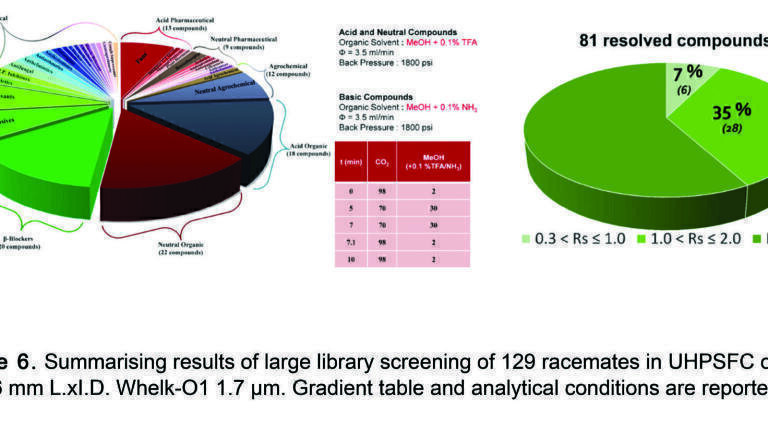
In order to demonstrate the potential of this CSP a large library screening was performed by Gasparrini’s group [21]. A previous version of the Whelk-O1 sub-2 µm was used, where the selector was bonded on 1.7 µm Thermo Syncronis silica and packed into a 50x4.6 mm. column (Figure 6). 129 chiral compounds of pharmaceutical interest where randomly collected (acidic, neutral and basic) and analysed in one working day: 81 out of 129 racemates were resolved under identical eluting conditions using a 9 min methanol in CO2 gradient with 0.1% TFA or 0.1% NH3 added for acidic and basic compounds, respectively. A flow rate of 3.5 mL/min allowed very fast and highly efficient separations, most of samples were eluted and successfully separated in less than 4 min. As expected, the best results were obtained concerning acid and neutral samples, but this CSP showed great results also on basic analytes: 47% of basic racemates were separated with a resolution value higher than 0.3, demonstrating the possibility to use this selector for acid, neutral and also basic chiral molecules.
A complete characterisation from the kinetic point of view of the UHPC-Whelk-O1 1.8 µm was performed and compared to those of the equivalent 3.5 and 5 µm commercial Whelk-O1 columns, with great results: efficiencies higher than 260,000 N/m on trans-stilbene oxide at a high optimal flow-rate of 1.8 mL/min were reached. The resulting separation was completed in less than 2 minutes with a reduction of the analysis time of more than four times in comparison with the 5 µm column. The UHPC-Whelk-O1 was also used in UHPSFC maintaining the same trend of UHPLC with very high efficiencies and very high optimal linear velocities. The separation of the two enantiomers of trans-stilbene oxide was completed in just 1 minute.
This CSP was packed into a 5cm long column and was used to screen 129 chiral compounds in sub-critical fluid chromatography with a 9-min screening method. 63% of this library was resolved in just ‘one working day’.
The authors would like to thank Professor Francesco Gasparrini for his support and for his invaluable advice, Waters s.p.a. (Milano, Italy) for providing UPC2 instrumentation and Dr E. Bianchini, Department of Chemistry and Pharmaceutical Sciences, University of Ferrara, for elemental analysis measurements.
[1] C.L. Barhate, M.F. Wahab, Z.S. Breitbach, D.S. Bell, D.W. Armstrong, Analytica Chimica Acta, 898 (2015) 128-137.
[2] D. Kotoni, A. Ciogli, C. Molinaro, I. D’Acquarica, J. Kocergin, T. Szczerba, H. Ritchie, C. Villani, F. Gasparrini, Anal. Chem. 84 (2012) 6805−6813.
[3] M. Lämmerhofer, W. Lindner, J. Chromatogr. A 741 (1996) 33−48.
[4] C.V. Hoffmann, R. Pell, M. Lämmerhofer, W. Lindner, Anal. Chem. 80 (2008) 8780−8789.
[5] D.W. Armstrong, Y. Tang, S. Chen, Y. Zhou, C. Bagwill, J.-R. Chen, Macrocyclic antibiotics as a new class of chiral selectors for liquid chromatography, Anal. Chem. 66 (1994) 1473−1484.
[6] A. Berthod, X.H. Chen, J.P. Kullman, D.W. Armstrong, F. Gasparrini, I. D’Acquarica, C. Villani, A. Carotti, Anal. Chem. 72 (2000) 1767−1780.
[7] I. D’Acquarica, F. Gasparrini, D.Misiti, M. Pierini, C. Villani, Adv. Chromatogr. 46 (2008) 109−173.
[8] O.H. Ismail, A. Ciogli, C. Villani, M. De Martino, M. Pierini, A. Cavazzini, D.S. Bell, F. Gasparrini, J. Chromatogr. A 1427 (2016) 55–68.
[9] D.W. Armstrong, Y. Liu, K.H. Ekborgott, Chirality 7 (1995) 474−497.
[10] W.H. Pirkle, J.M. Finn, J. Org. Chem. 46 (1981) 2935−2938.
[11] W.H. Pirkle, C.J. Welch, B. Lamm, J. Org. Chem. 57 (1992) 3854−3860.
[12] F. Gasparrini, D. Misiti, M. Pierini, C. Villani, J. Chromatogr. A 724 (1996) 79-90.
[13] S.G. Allenmark, S. Andersson, P. Möller, D. Sanchez, Chirality 7 (1995) 248−256.
[14] Y. Okamoto, M. Kawashima, K. Hatada, J.Chromatogr. A 363 (1986) 173−186.
[15] S. Allenmark, B. Bomgren, H.Borén, J. Chromatogr. A 264 (1983) 63−68.
[16] F. Gasparrini, D. Misiti, R. Rompietti, C. Villani, J. Chromatogr. A 1064 (2005) 25−38.
[17] D. Kotoni, C. Villani, D.S. Bell, D. Capitani, P. Campiglia, F. Gasparrini, J. Chromatogr. A 1297 (2013) 157−167
[18] G. Cancelliere, A. Ciogli, I. D’Acquarica, F. Gasparrini, J. Kocergin, D. Misiti, M. Pierini, H. Ritchie, P. Simone, C. Villani, J. Chromatogr. A 1217 (2010) 990−999.
[19] F. Ai, L. Li, S.-C. Ng, T.T.Y. Tan, J. Chromatogr. A 1217 (2010) 7502−7506.
[20] A. Cavazzini, N. Marchetti, R. Guzzinati, M. Pierini, A. Ciogli, D. Kotoni, I. D’Acquarica, C. Villani, F. Gasparrini, TrAC 63 (2014) 95−103.
[21] L. Sciascera, O. Ismail, A. Ciogli, D. Kotoni, A. Cavazzini, L. Botta, T. Szczerba, J. Kocergin, C. Villani, F. Gasparrini, J. Chromatogr. A 1383 (2015) 160−168.
[22] Y. Min, Z. Sui, Z. Liang, L. Zhang, Y. Zhang, J. Pharm. Biomed. Anal. 114 (2015) 247−253.
[23] D.C. Patel, Z.S. Breitbach, M.F. Wahab, C.L. Barhate, D.W. Armstrong, Anal. Chem., 87 (2015) 9137–9148
[24] O.H. Ismail, A. Ciogli, C. Villani, L. Pasti, A. Cavazzini, F. Gasparrini, S. Anderson, J. Kocergin, M. Catani, J. Chromatogr. A, (2016), in preparation.
[25] W.H. Pirkle, C.J. Welch, J. Liq. Chromatogr., 15 (1992), 1947
[26] D. Kotoni, A. Ciogli, I. D’Acquarica, J. Kocergin, T. Szczerba, H. Ritchie, C. Villani, F. Gasparrini, J. Chromatogr. A 1269 (2012) 226.
[27] T.A. Berger, J. Chromatogr. A, 1459 (2016) 136-144.
[28] F. Gritti, G. Guiochon, J. Chromatogr. A 1218 (2011) 4452–4461.
[29] A. Grand-Guillaume Perrenoud, J.-L. Veuthey, D. Guillarme, J. Chromatogr. A 1266 (2012) 158−167.
