-
 Figure 1: Charge variants of IgG4 based monoclonal antibodies can be successfully evaluated by AEX-MS.
Figure 1: Charge variants of IgG4 based monoclonal antibodies can be successfully evaluated by AEX-MS. -
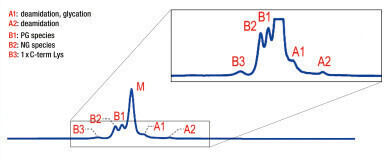 Figure 2: Native AEX-MS charge variant analysis, shown basic (B) and acidic (A) variant peaks as well as the main species (M) (5 μg mAb injection) [1].
Figure 2: Native AEX-MS charge variant analysis, shown basic (B) and acidic (A) variant peaks as well as the main species (M) (5 μg mAb injection) [1]. -
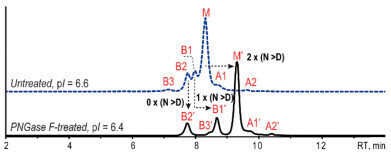 Figure 3: Native AEX-MS analysis before (blue) and after PNGase F-treatment (black) [1].
Figure 3: Native AEX-MS analysis before (blue) and after PNGase F-treatment (black) [1].
Liquid Chromatography
AEX-MS charge heterogeneity analysis of an IgG4-based mAb
Oct 24 2023
Cation exchange chromatography (CEX) is very well suited for characterising the charge heterogeneity of biomolecules. This also applies to most commercially available monoclonal antibodies (mAbs). They are often based on IgG1 and have a high isoelectric point (pI). Anion exchange chromatography (AEX), has so far only been used for relatively basic proteins.
IgG4-based mAbs are becoming increasingly important. This application note demonstrates that the charge heterogeneity of an IgG4-based mAb with a pI of 6.6 (proprietary) can be successfully evaluated using a strong anion exchange column (SAX), BioPro IEX QF, in conjunction with MS detection [1].
With this AEX method three basic as well as two acidic charge variants can be separated from the main species (see Figure 2). Furthermore, this method seems to be highly sensitive to the Fc N-glycosylation macroheterogeneity as the fully glycosylated (FG) main species is separated from the partially glycosylated (PG) peak B1 and the non-glycosylated (NG) species B2, which elute earlier. This leads to the assumption that the surface charge rather than the intrinsic charge is decisive for the AEX separation.
Improving resolution
mAbs with a lower pI are better separated in AEX mode. Therefore, the pI of the mAb was lowered by treatment with PNGase F-mediated deglycosylation. This reaction removes N-glycans and simultaneously converts the glycan-bearing asparagine (Asn) residue to aspartic acid (Asp), which all IgG4 mAbs contain in the Fc region in each of the two heavy chains. Therefore, up to two Asn to Asp conversions can be expected.
After the PNGase F-treatment the retention time of the FG main species and the PG species B1 is extended (see Figure 3) due to the increased acidity induced by now one respectively two Asp residues. The retention time of the NG species B2 remains unchanged. Due to lowering the pI from 6.6 to 6.4 the resolution is massively improved.
Fc critical quality attribute monitoring
This AEX-MS method can also be applied for subunit analysis of mAbs after IdeS digestion. The Fc fragments were treated with PNGase F-mediated glycosylation to profit from the improved resolution and further investigate the glycosylated species. For the Fc fragment analysis, a lower temperature of 25 °C shows improved peak shape and charge variant separation over the analysis at 45 °C (see full application note).
Four basic and two acidic variants can be identified. In addition to the NG B3 and the PG B1 peak, B2 is identified as a fully glycosylated species with one unprocessed C-term Lys while B4 is identified as a partially glycosylated species with one unprocessed C-term Lys. These findings were confirmed by peptide mapping.
Summary
AEX-MS analysis is a good alternative for the charge heterogeneity characterisation of IgG4-based mAbs. The resolution of the glycosylated variants can be further improved by PNGase F-mediated deglycosylation. AEX-MS methods are suitable for the Fc critical quality attribute monitoring of IgG4-based mAbs, while CEX remains the better option for F(ab’)2 subunit analysis.
Literature
[1] A. Liu, Y. Yan, S. Wang, N. Li, Anal. Chem. 2022, 94, 6355−62
Digital Edition
Chromatography Today - Buyers' Guide 2022
October 2023
In This Edition Modern & Practical Applications - Accelerating ADC Development with Mass Spectrometry - Implementing High-Resolution Ion Mobility into Peptide Mapping Workflows Chromatogr...
View all digital editions
Events
Nov 12 2024 Tel Aviv, Israel
Nov 18 2024 Shanghai, China
Nov 18 2024 Plainsboro, NJ, USA
Nov 20 2024 Karachi, Pakistan
Feb 03 2025 Dubai, UAE













