Affinity
Published over 7 years ago. See the latest and most current information on Affinity.
A new three-phase partitioning system is described in which a nanoparticulate analyte sequestering transport phase (ASTP) causes analytes to elute in the column void volume followed by impurities. The function of the ASTP is to structure specifically sequester an analyte (An) with high affinity and be of sufficient size to be excluded from a size exclusion chromatography (SEC) column. The linear velocity of non-analytes in contrast is retarded by diffusion into pores of the column packing during SEC. Analytes in the ASTP:An complex are detected by an analyte specific method. Fluorescent sandwich methods were used for detection in the examples shown.
Based on advances in mass spectrometry analysis of proteins and their peptide fragments it has been predicted that the human proteome of many organisms is composed of more than a million proteins, many of which are proteoforms of very similar structure [1]. Although gas phase analysis is very powerful and exciting, most of these proteins have never been ‘held in the hand’, so to speak. Total characterisation and analysis require protein purification. There is also the issue of cost and speed of analysis. Sample preparation can take hours with mass spectral methods [2]. There is a need for inexpensive methods that rapidly identify proteoforms in routine analyses. Millions of protein specific analyses are performed annually in clinical diagnostics [3]. With samples as complicated as blood, existing separation systems do not differentiate well between structural isoforms [1]. Improvements in protein separation methods are needed.
Liquid chromatography (LC) systems have been at the core of protein purification and structure analysis for more than 50 years. The underlying two-phase partitioning mechanism enabling protein resolution remains unchanged [4]. Although improvements in stationary phases, enhanced particle fabrication methods, and reductions in particle size have greatly advanced the resolution and speed of this separation mode, the peak capacity of most liquid chromatography columns is still a hundred or less. With biological extracts of 105 or more components there is a high probability individual peaks will contain many proteins.
The discussion below will focus on a new, inexpensive mobile affinity sorbent chromatography (MASC) approach that increases resolution and speed in routine analyses by incorporating a transport phase into conventional two-phase LC systems [5]. The major advantage of MASC is that it rapidly increases selectivity.
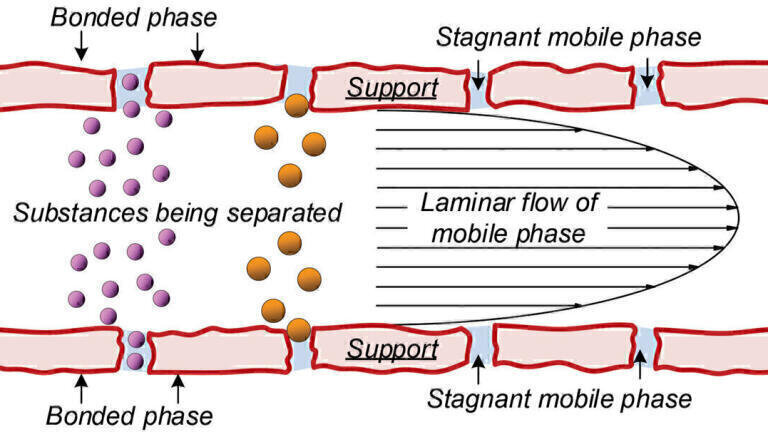
Proteins are separated in LC by repetitive differential partitioning between a stationary phase (PS) and mobile phase (Pm) [6]. This is achieved by Pm transport of mixtures through long, narrow channel networks bearing a stationary phase (PS) embedded in a packed particle bed. Substances spending time on a solid Ps or in stagnant Pm take longer to elute. Stagnant Pm is located within porous, insoluble supporting matrices (Figure 1).
With size exclusion chromatography (SEC) stagnant Pm is the stationary phase [9]. Small substances diffuse more easily and completely into stagnant Pm within pore matrices than large molecules. The longitudinal velocity of a protein or peptide is zero while inside pores. This allows substances in the Pm to move ahead of those in pores. In contrast, larger proteins are partially or totally exclude from pore matrices. This means they spend more time in the mobile phase and move through the system faster.
The idea that Pm moves through a particle bed in a laminar flow profile is derived from hydrodynamic chromatography (HDC). Early HDC studies showed that long packed columns separate colloids of varying size [7]. Larger analytes elute first and smaller ones later, due to preferential sampling of the streamlines of flow in the interstitial medium of the packed column [8]. Because all analyte sequestering transport particles are of the same size and columns are short HDC plays a minimal role in MASC resolution.
The theory behind three-phase separation systems comes from literature describing the immobilisation of proteins on the surface of 50 - 100 nm nanoparticles [5,10] to create megadalton (mDa) size immune complexes that remain suspended in solution and retain their biologically activity [11]. In the chromatography application described below these particles will be referred to as an analyte sequestering transport phase (ASTP). Association of an antigen (An) with an ASTP bearing an antibody is represented
in the reaction
The association constant (Ka) for complex formation is expressed by the equation
where CASTP and CAn are the concentrations of ASTP and An, respectively and CASTP:An is the concentration of the ASTP:An complex. The rates of association and dissociation are represented by the symbols ka and kd, respectively. Ideally Ka and the ka/kd ratio will be 106 or higher. An association constant of this magnitude allows the ASTP:An complex to be transported through a chromatography column without dissociation.
The analytical significance of this is threefold. One is that ASTP:An complexes are soluble in aqueous Pm and sufficiently small to be transported through a chromatography column. This allows a sorbent particle to be used as a transport phase as opposed to insoluble stationary phases. Obviously an ASTP cannot be used as a transport phase in the remote case where it binds to a very large, multiple protein complex [12] and precipitates. The very high binding affinity of analytes for an ASTP is a second asset. Analyte association with Ps is precluded. The fact that non-analytes (sample impurities) do not associate with an ASTP is still another advantage. This makes it possible to rapidly differentiate between analytes and impurities. The final asset of ASTP:An complex formation is that upon binding an analyte will become very different from impurities of very similar structure (Figure 2). When in an ASTP:An complex the effective molecular weight of an ASTP sequestered analyte is 5X to 10X larger than that of protein impurities.
Following analyte sequestration the issue is how to separate and detect an ASTP:An complex relative to impurities. By design these complexes are soluble in common aqueous mobile phases and of a size amenable to separation by SEC (Figure 2). With an SEC column having 30 nm pores a megadalton size ASTP:An complex will elute in the void volume while impurities will be retained by Ps and elute within a single column volume. Short columns could be eluted at high flow rates within 60 - 120 sec. Few impurities would coelute with the ASTP:An complex based on the huge difference in their relative size.
The three phase system described above would allow three layers of selectivity (Figure 2). The first is the very high selectivity of affinity selectors such as antibodies and aptamers. Antibodies can even differentiate between proteoforms as known with isoenzymes [13] and post-translational modification variants of proteins [14]. The second is the very large shift in the effective size of an analyte when it binds to an ASTP. The significance of this is that subsequent to binding small analytes elute in the SEC void volume irrespective of their size. The third is the selectivity of the SEC column in size separating the ASTP:analyte complex from impurities.
Theoretically three-phase chromatography could be carried out in two ways. One would be by continuous addition of ASTP to the SEC mobile phase. In this mode the ASTP:An complex would be formed in the column. The concern with this approach is that large amounts of expensive ASTP would be consumed in the absence of An. An alternative would be a zonal mode. In this mode the ASTP:An complex could be formed in a sample vial and then injected into the SEC column. Because the association constant Ka is at least 106 these two modes will be equivalent in terms of resolution. This allowed all experiments described herein to be carried out in the zonal elution mode to reduce ASTP cost.
Critical elements in ASTP selection and fabrication are that i) the nanoparticles be hydrophilic and as nearly neutral as possible to preclude aggregation and non-specific binding of sample impurities, ii) the ASTP Core material should be amenable to covalent immobilisation of proteins, iii) the ASTP should be of 50-100 nm in size to assure exclusion from pores, and iv) an ASTP should have minimal impact on mobile phase viscosity. Two types of material were evaluated, natural polysaccharides and synthetic inorganic particles. Both worked well, but in the end, very high molecular weight dextran was found to have the fewest limitations. Dextrans used in this work exceed 1 mDa and were natural products of Leuconostoc mesenteroides [15].
Covalent linkage of proteins to dextran core particles was achieved in two ways. One was by oxidative cleavage of vicinal diol residues with periodate to form aldehydes [16]. Amine groups in proteins were coupled to the aldehyde rich dextan by Schiff base formation followed by NaCNBH3 reduction to a secondary amine linkage [17]. A problem with this approach is extensive hemiacetal formation in the oxidized dextran that makes it difficult to control the number of aldehyde residues available for protein coupling [18].
The second approach was to carboxymethylate the dextran. This was achieved by the addition of chloroacetic acid in 8M KOH [19]. Proteins were amide coupled to the CM-dextran via hydroxysuccinimide assisted water soluble carbodiimide catalysis [20]. This method of ASTP formation was preferred in that it was easier to control the number of functional groups available for protein immobilisation.
The above three-phase model is the mechanistic basis for mobile affinity sorbent chromatography (MASC). Validation of MASC was achieved in two ways. The first was to show that an ASTP with an immobilised affinity selector would bind a protein and cause it to elute in the void volume of an SEC column (Figure 3). Dextran based ASTP with covalently linked protein A/G (ASTP~PA/G) was added to a sample bearing an excess of fluorescently labelled IgG - [IgG*; the antibody is covalently labelled with fluorescein isothiocyanate (FITC)]. Protein A/G is a recombinant protein hybrid that contains domains of both protein A and protein G, causing it to bind to the Fc region of most mammalian antibodies [21]. The ASTP~PA/G :IgG* complex was subsequently formed and injected into a 4.6 × 300 mm, 5 μm particle diameter, 30 nm pore diameter Sepax SEC column (Figure 3). IgG* was detection by fluorescence at excitation and emission wavelengths of 494 nm and 510 nm, respectively. As judged from the elution of protein standards in the accompanying chromatogram the fluorescence peak eluting at 2.1 min in the void volume clearly contains IgG* transported through the column without entering the pores of the SEC column. Unbound IgG* elutes at 3.2 min. This experiment demonstrated that (i) protein A/G retained IgG binding specificity when bound to the dextran based ASTP nanoparticle, (ii) analyte IgG* could be isocratically separated from other protein species using a single column volume of mobile phase in the MASC mode, (iii) ASTP~PA/G bound IgG* behaves as if it had a molecular weight in the range of 1 mDa, and iv) analyte elution behaviour was independent of analyte molecular weight.
There is concern when producing or buying an antibody that it retains antigen affinity. The experiments described below addressed that issue. A saturating concentration of fluorescent labelled secondary antibody (2AbmIgG*) targeting mouse IgG was added to mouse 1Abfitc solutions of the same volume that varied in concentration. It is assumed in this experiment that 2AbmIgG* will bind to both active and inactive forms of the fluorescein isothiocyanate (FITC) targeting antibody. Samples were incubated for 30 min to allow formation of the 1Abfitc: 2AbmIgG* complex and then added to 100 μL of human plasma bearing ASTP~FITC. Following incubation for at least another 30 min to allow formation of the ASTP~FITC: 1Abfitc: 2AbmIgG* complex samples were examined by MASC (Figure 4). The Sepax SEC column used in these experiments was the same as in Figure 3.
The peak in column void volume at 2.1 min is clearly the ASTP~FITC: 1Abfitc: 2AbmIgG* complex while that at 3.2 min is unassociated mouse targeting secondary antibody 2AbmIgG*. The very small peak eluting at 2.6 min can either be a dimer of 2AbmIgG* or 1Abfitc: 2AbmIgG*. If it is 1Abfitc: 2AbmIgG* the primary antibody no longer recognises FITC antigen.
Mobile affinity sorbent chromatography (MASC) will be most useful in routine separations involving small numbers of analytes. The most powerful feature of the method is the simultaneous use of two partitioning phases that cause analytes of interest to elute in the column void volume ahead of impurities. One of these phases is a nanoparticulate transport phase of ~2 mDa that i) binds analytes with high affinity and selectivity based on their 3D structure, ii) causes analyte to elute from columns ahead of impurities, and iii) precludes interactions of analytes with the column stationary phase (Ps). Ps in contrast i) retards the linear velocity of non-analytes (impurities), ii) is prevented from interacting with analytes, and iii) has no impact on analyte retention.
When MASC separations are carried out with a size exclusion column i) the entire analysis of a sample, irrespective of sample complexity can be achieved with a single mobile phase volume, ii) separations are achieved by isocratic elution, iii) column recycling is unnecessary, and iv) analyte carryover is precluded by using new analyte sequestering transport phase in each analysis.
A limitation of MASC is in distinguishing between analytes and transport particles at the point of detection. Analytes and an ASTP can have indistinguishable absorbance properties. That problem was addressed herein via a fluorescent sandwich format much as in sandwich type immunological assays. Detection of the FITC targeting antibody 1Abfitc for example was achieved by creating the fluorescent ASTP~FITC: 1Abfitc: 2AbmIgG* sandwich. This approach has the advantage of adding yet another level of selectivity during detection but increases the analytical complexity and cost of the analysis.
The authors gratefully acknowledge support of this work through an NIH Phase II Phase II SBIR grant (5R44GM116663) entitled ‘Sample Preparation; The Achilles Heel of Mass Spectrometry Based Diagnostics II’.
1. Regnier, Fred E.; Kim, JinHee. Proteins and Proteoforms: New Separation Challenges. Anal. Chem. (2018), 90, 361−373.
2. Klont, Frank; Bras, Linda; Wolters, Justina C.; Ongay, Sara; Bischoff, Rainer; Halmos, Gyorgy B.; Horvatovich, Péter. Assessment of Sample Preparation Bias in Mass Spectrometry-Based Proteomics. Anal. Chem., (2018), 90 (8), pp 5405–5413.
3. Gregorich, Zachery R.; Ge, Ying.Top-down proteomics in health and disease: Challenges and opportunities. Proteomics (2014), 14, 1195–1210.
4. McCue, Justin T.). Theory and use of hydrophobic interaction chromatography in protein purification applications. Meth. Enzymol. (2009), 463, 405–414.
5. Li, ZhiYu; Kim, JinHee; Regnier, Fred E. Mobile Affinity Sorbent Chromatography. Anal. Chem. (2017), 35(8), 510-511.
6. Meyer, Veronika R. Practical High-Performance Liquid Chromatography. (2010), 5th Edition, 344.
7. Small, H. Hydrodynamic chromatography a technique for size analysis of colloidal particles. J. Colloid Interface Sci. (1974), 48,147-161.
8. Striegel, A.M. Hydrodynamic chromatography: packed columns, multiple detectors, and microcapillaries. Anal. Bioanal. Chem. (2012), 402 (1), 77–81.
9. Fekete, S.; Beck, A.; Veuthey, J.-Luc; Guillarme, D. Theory and practice of size exclusion chromatography for the analysis of protein aggregates. J. Pharm. Biomed. Anal. (2014), 101, 161-173.
10. Schneck, N.A.; Lowenthal, M.; Phinney, K.; Lee, Sang B. Current trends in magnetic particle enrichment for mass spectrometry-based analysis of cardiovascular protein biomarkers. Nanomedicine. (2015), 10(3), 433–446.
11. Jana, N.R.; Earhart, C.; Ying, J.Y. Synthesis of Water-Soluble and Functionalized Nanoparticles by Silica Coating. Chem. Mater. (2007), 19 (21), 5074–5082.
12. Gao, Q.; Madian, A.G.; Liu, X.; Adamec, J.; Regnier, F.E. Coupling protein complex analysis to peptide based proteomics. J. Chromatogr., A (2010), 1217(49), 7661-7668.
13. Suzuki, T.; Tomita, K.; Murachi, T. Creatine kinase-inhibiting monoclonal antibodies: preparation and characterization of porcine MM isoenzyme-specific antibodies. Molec. Cell. Probes. (1988), 2(9), 157-167.
14. Elfineh, L; Classon, C; Asplund, A; Pettersson, U; Kamali-Moghaddam, M; Lind, S.B. Tyrosine phosphorylation profiling via in situ proximity ligation assay. BMC Cancer. (2014),14, 435.
15. Vettori, M.H.P.B.; Franchetti, S.M.M.; Contiero, J. Structural characterization of a new dextran with a low degree of branching produced by Leuconostoc mesenteroides FT045B dextransucrase. Carbohydrate Polymers. (2012), 88,1440–1444.
16. Betancor, L.; López-Gallego, F.; Hidalgo, A.; Alonso-Morales, N.; Fuentes, M.; Fernández-Lafuente, R.; Guisán; J.M. Prevention of interfacial inactivation of enzymes by coating the enzyme surface with dextran-aldehyde. J. Biotech. (2004), 110, 201–207.
17. Jia, Y.; Li, J. Molecular Assembly of Schiff Base Interactions: Const. Applic. Chem. Rev. (2015), 115, 1597−1621.
18. Evangelista, R. A.; Chen, F-T. A.; Guttman, A. Reductive amination of N-linked oligosaccharides using organic acid catalysts. J. Chromatogr. A. (1996), 745, 273-280.
19. Zhang R.; Tang M.; Bowyer A.; Eisenthal R.; Hubble J. Synthesis and characterization of a D-glucose sensitive hydrogel based on CM-dextran and concanavalin A. React. Funct. Polymers. (2006), 66, 757–767.
20. Chou, P.-H.; Chen, S.-H.; Liao, H.-K.; Lin, P.-C.; Her, G.-R.; Lai, A.C.-Y.; Chen, J.-H.; Lin, C.-C.; Chen, Y.J. Nanoprobe-Based Affinity Mass Spectrometry for Selected Protein Profiling in Human Plasma. Anal. Chem. (2005), 77, 5990-5997.
21. Hermanson, G.T. Immobilization of ligands on chromatography supports. Bioconjug. Tech. (Third Edition), (2013), 127-128.
